Are Oral Peptides Effective? The Science of Pills, Capsules, and Bioavailability
Written bySpartan Research Team
Can you take peptides orally? For pharmaceutical research, this question is not about consumer convenience. Instead, it’s about understanding how a peptide’s molecular structure, gastrointestinal physiology, and bioavailability interact.
Oral peptide research is a focal point in drug delivery science. Scientists are investigating how to overcome the biological barriers that limit effective absorption.
Peptide bioavailability remains a central challenge in pharmaceutical in
vestigations. This is mainly because of enzymatic degradation, pH instability, and first-pass metabolism.
This article examines the latest research findings, delivery technologies, and experimental methodologies. The emphasis is on laboratory and pharmaceutical development contexts.
To ensure reproducible results across laboratories, research requires high-purity, research-grade peptides with consistent specifications.
Spartan Peptides meets this critical need by providing rigorously quality-controlled peptides with comprehensive analytical documentation.
The science of oral peptide delivery
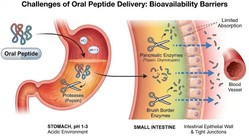
Oral peptide delivery has long been a significant challenge in pharmaceutical research. Peptides are large, hydrophilic macromolecules that face many obstacles in the gastrointestinal tract.
In research models, the barriers that reduce systemic bioavailability can be categorized into the following factors:
- Chemical
- Enzymatic
- Physiological
Gastrointestinal tract barriers in research
Research models show that orally administered peptides encounter a highly variable pH environment. This ranges from acidic gastric fluid to near-neutral intestinal conditions.
Acidic conditions can denature certain peptide structures. Alkaline conditions in the small intestine may alter its folding and charge distribution.
For peptides exceeding ~500 Da in molecular weight, gastrointestinal epithelial layers present a barrier to absorption.
Enzymatic degradation pathways
Peptide bioavailability research has consistently identified proteolytic degradation as a common cause of low oral absorption.
Peptidases in the stomach and intestine quickly hydrolyze peptide bonds, reducing the fraction of intact molecules available for systemic uptake.
pH stability in oral delivery research
Many peptides have a narrow pH tolerance, which can lead to faster degradation outside their optimal range. Even when molecules reach the portal circulation intact, first-pass hepatic metabolism can further reduce systemic concentrations.
In vitro and in vivo studies suggest that most oral peptides rely on paracellular diffusion or endocytosis for intestinal uptake. These processes are inherently inefficient for large, hydrophilic molecules.
Research approaches to enhance oral delivery
Pharmaceutical peptide studies have explored several strategies to improve oral peptide delivery:
- Permeation enhancers to transiently open tight junctions
- Enzyme inhibitors to protect against proteolysis
- Enteric coatings to bypass acidic gastric environments
- Nanoparticle encapsulation to improve stability and targeting
Bioavailability challenges in research studies
Peptide bioavailability studies consistently show that oral delivery significantly lowers systemic exposure compared to parenteral routes.
Absolute bioavailability of unmodified peptides often falls below 1%, primarily due to enzymatic degradation and limited epithelial permeability.
Comparative delivery research
Pharmaceutical research frequently compares oral and injectable administration. Injections offer nearly complete bioavailability, while peptides taken by mouth display negligible plasma concentrations. These findings call for delivery-enhancement strategies.
Factors affecting absorption
Variables influencing absorption in oral peptide research include:
- Peptide molecular weight and structural conformation: Larger peptides (>1000 Da) show dramatically reduced permeability coefficients. Secondary structures like beta-sheets can influence membrane interaction and enzymatic susceptibility.
- Stability across gastrointestinal pH gradients: pH transitions from stomach (pH 1-3) to duodenum (pH 6-7) cause conformational changes in many therapeutic peptides. Some sequences aggregate under acidic conditions, while others undergo irreversible denaturation.
- Interaction with dietary components: Food matrices can bind peptides through electrostatic or hydrophobic interactions. This reduces the available fraction for absorption.
- Co-administration with enzyme inhibitors or permeation enhancers: Protease inhibitors can improve stability. Sodium caprate and other fatty acid derivatives temporarily disrupt tight junctions, enhancing paracellular transport.
Methodologies for measuring bioavailability
Scientists employ multiple techniques to quantify oral peptide absorption:
- Pharmacokinetic profiling via plasma concentration-time curves: Serial blood sampling is used to calculate key parameters, like AUC, Cmax, and Tmax, which are essential for assessing bioavailability.
- Mass spectrometry for detecting intact peptide sequences: LC-MS/MS provides specificity to distinguish parent compounds from metabolites. This is critical for understanding degradation patterns.
- In vitro permeability assays using Caco-2 monolayers: These human intestinal cell models predict passive diffusion and active transport, although they lack mucous layers present in vivo.
- Animal model tracking with radiolabeled peptides: Isotopic labeling enables whole-body distribution studies. This technique also allows for the precise quantification of absorption efficiency.
Pharmaceutical research on oral formulation technologies
Advances in oral peptide formulation research are addressing stability and permeability challenges that have limited therapeutic applications.
Pharmaceutical peptide research has evolved from simply identifying barriers to developing sophisticated delivery systems.
Enteric coating technologies
Drug delivery research has developed advanced enteric polymers specifically engineered for peptide protection.
These pH-responsive coatings prevent the breakdown of sensitive molecules by gastric acid. This ensures controlled release in the neutral environment of the small intestine.
Nanoparticle and liposomal delivery systems
Lipid-based nanoparticles are a major focus in current pharmaceutical peptide research. These carriers encapsulate peptides in protective lipid bilayers, preventing enzymatic degradation while enhancing mucosal adhesion.
Liposomal encapsulation studies indicate improved stability profiles. Some formulations achieve 5-10 times the bioavailability improvements in unprotected peptides.
Permeation enhancers and protease inhibitors
Permeation enhancer research has identified compounds, like sodium caprate, that transiently open epithelial tight junctions. Protease inhibitor studies demonstrate that co-formulation with aprotinin significantly reduces enzymatic degradation.
These strategies are particularly critical for ensuring the bioavailability of compounds like BPC-157, a stable peptide currently being researched for its regenerative properties.
Cyclodextrin complexation research shows that inclusion complexes improve peptide solubility while protecting against hydrolysis.
Research methodologies for studying oral peptides
Evaluating oral peptide delivery systems requires sophisticated pharmaceutical research protocols that reconcile analytical precision and experimental reproducibility.
Modern peptide research methods provide a comprehensive assessment of absorption mechanisms, stability profiles, and systemic exposure.
In vitro dissolution testing
Laboratory dissolution studies employ standardized simulated gastric and intestinal fluids to evaluate peptide stability under physiological conditions.
Caco-2 and HT29-MTX cell monolayers serve as primary models for intestinal epithelial transport studies, measuring apparent permeability coefficients and transport mechanisms.
Animal model protocols and analytical methods
Pharmaceutical research protocols use rodent and non-human primate models for a comprehensive bioavailability assessment. Crossover study designs with wash-out periods enable comparative evaluation between oral and intravenous administration routes.
High-performance liquid chromatography coupled with tandem mass spectrometry represents the analytical gold standard for quantifying intact peptide concentrations in biological matrices.
Current research findings and clinical studies
Oral peptide clinical research has shown significant progress in overcoming traditional bioavailability barriers, with several therapeutic classes showing promising results.
Breakthrough formulations are advancing toward regulatory approval, while most programs remain in early-phase development.
Published research studies
GLP-1 Sema represents the most successful oral peptide clinical research achievement to date.
Sodium N-(8-[2-hydroxybenzoyl]amino)caprylate (SNAC) is an absorption enhancer with potential. This allows the formulation to achieve approximately 0.4–1% bioavailability compared to subcutaneous administration.
Pharmaceutical development programs are expanding this technology platform to other therapeutic targets, including parathyroid hormone analogues for osteoporosis treatment.
Comparative effectiveness and limitations
Head-to-head studies between oral and injectable peptide formulations reveal important trade-offs.
Oral dosing improves patient adherence, while injectables have better bioavailability. Oral formulations often require more doses to achieve comparable systemic exposure.
Current research faces methodological challenges, as inter-subject variability in absorption can exceed 200%.
Factors affecting oral peptide research success
Understanding oral peptide effectiveness factors is critical for designing robust studies. Research success depends on multiple interconnected variables that must be evaluated during the formulation stage.
Molecular characteristics and structural considerations
Peptide research variables demonstrate that molecular weight directly correlates with absorption efficiency.
Peptides exceeding 1000 Da show dramatically reduced permeability, while compounds below 500 Da achieve measurably higher bioavailability.
Basic residues, like lysine and arginine, create preferential cleavage sites for trypsin. Aromatic residues may enhance membrane interactions but increase total potential.
Most therapeutic peptides have a narrow pH tolerance. They are typically stable between pH 6–8, so researchers must account for the wide pH fluctuations in the gastrointestinal tract.
Food effect studies and dosing optimization
Research protocols demonstrate that concurrent food intake alters gastric emptying patterns and pH buffering capacity, significantly impacting absorption kinetics. Fed-state conditions can reduce bioavailability for many peptides.
Animal model studies reveal substantial individual variation in peptide absorption, with coefficients of variation often exceeding 50%.
Future directions in oral peptide research
Future peptide research integrates cutting-edge technologies and traditional pharmaceutical science to create multi-modal delivery platforms.
Pharmaceutical development trends indicate substantial industry investment in next-generation approaches.
Advanced nanotechnology and delivery innovations
Oral delivery innovations in nanotechnology focus on smart, responsive delivery systems. Polymeric nanoparticles with pH-triggered release mechanisms are being engineered for targeted intestinal uptake.
Studies use computational design to create inherently stable peptide analogues through amino acid substitutions and peptide cyclization.
Genetic engineering and peptide optimization
Future peptide research uses computational design to create stable peptide analogues. This is done by replacing cleavage-sensitive sequences with resistant alternatives while maintaining biological activity.
Peptide cyclization research shows promise for creating conformationally constrained molecules with enhanced stability and membrane permeability.
Artificial intelligence and regulatory landscape
Machine learning algorithms are streamlining formulation development by predicting optimal excipient combinations and dosing regimens.
AI-driven platforms analyze vast datasets, from dissolution testing and clinical outcomes, to identify unrecognized patterns. Regulatory agencies are developing streamlined pathways for complex oral peptide formulations, recognizing their therapeutic potential.
Pharmaceutical development trends point toward personalized peptide therapy, where genetic markers predict individual absorption capacity and optimal formulation selection.
At Spartan Peptides, our mission is to advance the peptide science field and offer products that make a real difference in people’s lives. We are dedicated to offering products with the highest standards of purity to support cutting-edge research and innovation.
Research applications and study design considerations
Designing robust oral peptide studies requires established pharmaceutical research methods for data integrity and regulatory compliance.
Peptide research design must reconcile rigor and feasibility to address the unique challenges of low-bioavailability compounds.
Study design and control considerations
Bioavailability study protocol development requires carefully matched positive and negative controls.
Researchers typically include intravenous administration as a bioavailability reference and vehicle-only controls for baseline measurements. Power analysis for oral peptide studies must account for high coefficients of variation.
For regulatory acceptability, pharmaceutical research methods must include comprehensive analytical validation and Good Laboratory Practice (GLP) compliance.
Ethical and regulatory compliance
Research protocols must meet institutional review board requirements and animal welfare guidelines. Documentation standards must support future regulatory submissions.
Advancing the science of oral peptide therapeutics
Oral peptide research continues to advance despite significant bioavailability challenges. Formulation technologies and delivery systems are showing measurable progress in overcoming enzymatic degradation and absorption barriers.
Pharmaceutical research and development efforts are yielding promising results through nanotechnology applications, AI-driven optimization, and sophisticated enhancement strategies, though most remain in preclinical stages.
Integrating advanced analytical methods, innovative formulation approaches, and emerging technologies positions the field for substantial breakthroughs in pharmaceutical development.
Ready to learn more about oral peptides? Spartan Peptides offers a comprehensive catalog of high-quality peptides to advance your oral peptide delivery research.
Frequently Asked Questions About Oral Peptide Bioavailability
Q: Why are most research peptides not administered orally?
A: Most peptides are rapidly degraded in the gastrointestinal tract by proteolytic enzymes (proteases and peptidases) before reaching systemic circulation. The stomach’s acidic environment and intestinal enzymes break peptide bonds efficiently. Additionally, the tight junctions of the intestinal epithelium present a significant absorption barrier for hydrophilic molecules larger than approximately 500 daltons. Research indicates this limits oral bioavailability of most peptides to very low levels.
Q: Are there any peptides that show meaningful oral bioavailability?
A: Some research suggests certain small peptides or specially formulated peptides can achieve meaningful oral bioavailability. BPC-157 has shown interesting activity via oral administration in animal studies, potentially through local gut action rather than systemic absorption. Cyclic peptides and those with unusual structural features may also resist proteolytic degradation. Oral peptide delivery is an active area of pharmaceutical research.
Q: What strategies are researchers investigating to improve oral peptide delivery?
A: Current research approaches include nanoparticle encapsulation, lipid nanoparticles, mucoadhesive delivery systems, PEGylation to increase molecular stability, protease inhibitor co-administration, and peptide cyclization. Pharmaceutical companies have invested significantly in these approaches, as oral delivery would dramatically improve patient compliance for peptide-based therapeutics.
Q: How does nasal administration compare to oral for peptide research?
A: Nasal (intranasal) administration bypasses much of the GI degradation problem by delivering peptides directly to the highly vascularized nasal mucosa. Some peptides, particularly nootropics like Semax and Selank, are specifically researched via nasal delivery due to the potential for direct transport along olfactory nerves to the central nervous system, bypassing the blood-brain barrier.
Q: What analytical methods do researchers use to study peptide bioavailability?
A: Common methods include plasma concentration monitoring via LC-MS/MS (liquid chromatography-mass spectrometry), radiolabeling studies to track peptide distribution, receptor binding assays to assess functional activity at target sites, and pharmacokinetic modeling to compare absorption rates across different administration routes. These techniques help establish whether measured effects reflect systemic absorption or local activity.
Related Research Resources
- How to Reconstitute Peptides: Step-by-Step Guide
- Semax product (intranasal nootropic peptide)
- Are Peptides Legal? Legal Landscape
- Browse all research peptides
Ready to advance your Research Peptides research?
Spartan Peptides offers ≥98% purity research peptides with full in-house purity verification and in-house quality testing.
⚠️ Research Use Only — Not for Human Consumption
The peptides discussed in this article are intended for laboratory and research purposes only. They are not intended for human consumption. All information presented is based on published preclinical research and is provided for educational purposes only.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →