CJC-1295 + Ipamorelin: The Complete 2026 Research Guide
Written bySpartan Research Team
In the expanding landscape of growth hormone research, few combinations have attracted as much scientific interest as CJC-1295 and Ipamorelin. This synergistic peptide stack operates through two distinct but complementary receptor pathways, producing amplified growth hormone (GH) pulses that far exceed what either compound achieves independently.

Whether you’re conducting preclinical research on metabolic regulation, body composition modulation, or GH axis dynamics, understanding the precise pharmacology of CJC-1295 + Ipamorelin is foundational. This guide synthesizes the current body of research, compares this stack to alternatives, and outlines practical protocols for laboratory use.
For broader context on how peptides function at the molecular level, see our Complete 2026 Peptide Research Guide. For general principles of synergistic combinations, visit our Peptide Stacking Research Guide.
What Is CJC-1295? The GHRH Analog Explained
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), the endogenous 44-amino-acid neuropeptide produced in the hypothalamus. In physiological conditions, GHRH travels to the anterior pituitary via the hypothalamic-pituitary portal system and binds GHRH receptors (GHRHR), triggering GH synthesis and secretion.
The original native GHRH has a plasma half-life of less than 10 minutes due to rapid degradation by dipeptidyl peptidase-IV (DPP-IV) and other proteases. CJC-1295 was engineered to overcome this limitation through two key structural modifications:
- Substitution at positions 2, 8, 15, and 27 to confer DPP-IV resistance
- Addition of a lysine linker at the C-terminus for optional conjugation with Drug Affinity Complex (DAC)
DAC vs. No-DAC: What the Research Shows
CJC-1295 exists in two primary research forms, and the distinction matters significantly for experimental design:
CJC-1295 with DAC (also called DAC:GRF) incorporates a maleimidoproprionic acid (MPA) moiety that forms a covalent bond with serum albumin after injection. This albumin-binding dramatically extends the half-life to 6–8 days in human studies (Ionescu & Frohman, 2006, PMID: 16682504). The result is near-continuous GHRH receptor stimulation — a “GH bleed” rather than pulsatile secretion.
CJC-1295 without DAC (also called Modified GRF 1-29 or Mod GRF 1-29) retains the DPP-IV resistant backbone without the albumin-binding chemistry. Its half-life is approximately 30 minutes, producing discrete, pulsatile GH release that more closely mirrors physiological rhythms. This is the form typically studied in combination with Ipamorelin.
Most stacking research uses CJC-1295 without DAC because pulsatile GH release is considered closer to endogenous patterns and avoids potential receptor desensitization associated with chronic, non-pulsatile stimulation.
What Is Ipamorelin? The Selective Growth Hormone Secretagogue
Ipamorelin (NNC 26-0161) is a pentapeptide growth hormone secretagogue receptor (GHSR) agonist developed by Novo Nordisk in the late 1990s. It belongs to the GH secretagogue family — synthetic compounds that mimic ghrelin’s ability to stimulate GH release through a receptor pathway entirely separate from GHRH.
The GHSR (also known as the ghrelin receptor) is a G-protein coupled receptor expressed in the pituitary, hypothalamus, and peripheral tissues. When activated, GHSR stimulates somatotrophs through phospholipase C and intracellular calcium signaling — a mechanism distinct from the cAMP pathway activated by GHRHR.
Why Ipamorelin Stands Out Among GH Secretagogues
Research interest in Ipamorelin intensified due to its remarkable receptor selectivity. Early GH secretagogues like GHRP-6 and GHRP-2 stimulated GHSR but also caused significant release of cortisol, prolactin, and ACTH — hormones that can confound research outcomes and create unwanted variables.
Ipamorelin, by contrast, demonstrates minimal off-target activity. A landmark study by Raun et al. (1998, PMID: 9665836) published in European Journal of Endocrinology demonstrated that Ipamorelin produced robust GH release in rats comparable to GHRP-6, but with significantly lower cortisol and ACTH responses. This selectivity profile makes it highly suitable for clean preclinical experimental designs.
Key pharmacological properties of Ipamorelin:
- Plasma half-life: Approximately 2 hours
- Receptor: GHSR-1a (ghrelin receptor)
- Off-target hormones: Minimal cortisol, prolactin, ACTH stimulation
- GH release pattern: Discrete, pulsatile
How CJC-1295 and Ipamorelin Work Together: Synergistic GH Pulse Amplification
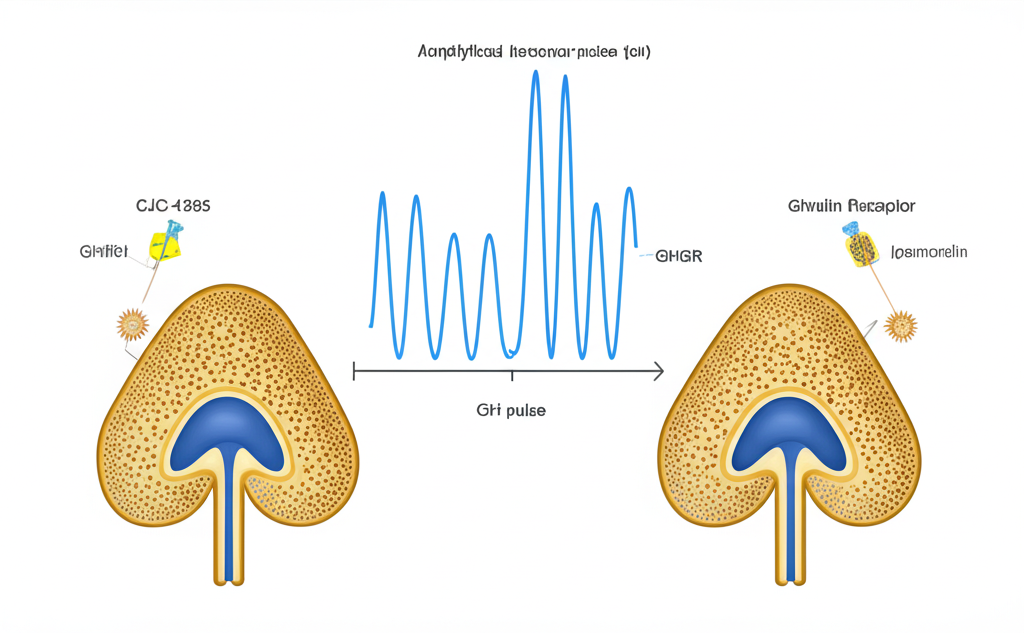
The scientific rationale for combining CJC-1295 with Ipamorelin lies in their complementary receptor mechanisms. The pituitary somatotroph has distinct receptor populations for GHRH (GHRHR) and ghrelin (GHSR-1a). When both are activated simultaneously, the intracellular signaling cascades interact synergistically:
- CJC-1295 activates GHRHR → increases intracellular cAMP → activates protein kinase A → enhances GH gene transcription and vesicle exocytosis
- Ipamorelin activates GHSR-1a → activates phospholipase C → mobilizes intracellular calcium → triggers additional GH vesicle fusion
These two pathways converge at the somatotroph and produce a supraphysiological GH pulse that neither compound achieves alone. Research in porcine models (Jørgensen et al., 2001, PMID: 11420165) demonstrated that co-administration of a GHRH analog with a GHSR agonist produced GH responses 2–4 times greater than either compound at equivalent doses.
Additionally, Ipamorelin suppresses somatostatin — the endogenous GH-inhibiting hormone — while CJC-1295 promotes GH synthesis. This dual action on both the accelerator (GHRH pathway) and the brake (somatostatin suppression) creates conditions for maximal GH release within each pulse.
The combined effect is a GH secretion profile characterized by:
- Higher peak amplitude per pulse vs. either compound alone
- Preserved pulsatility — physiologically relevant pattern retained
- Downstream IGF-1 elevation — liver responds to GH pulses with IGF-1 synthesis
- Minimal somatostatin interference — release window maximized
For researchers interested in the broader evidence base for growth hormone peptides, see our dedicated analysis: Do Growth Hormone Peptides Really Work?
Research Findings: Key Studies on CJC-1295 + Ipamorelin
The scientific literature supporting CJC-1295 Ipamorelin research spans preclinical animal models and human phase I/II trials. Below is a synthesis of key findings:
CJC-1295 Clinical Evidence
Ionescu & Frohman (2006) — The foundational human study of CJC-1295 (PMID: 16682504). In a dose-escalation study of 21 healthy adults, subcutaneous CJC-1295 (with DAC) at doses of 30–120 mcg/kg produced dose-dependent increases in mean GH concentrations up to 10-fold above baseline, with sustained elevation for 6+ days. IGF-1 levels increased 1.5–3-fold over the 7-day observation window.
Teichman et al. (2006) — Follow-up study (PMID: 16822944) examining multiple CJC-1295 doses in healthy adults. Confirmed prolonged GH and IGF-1 elevation with favorable tolerability profile. No significant changes in cortisol, thyroid hormones, or glucose were observed.
Ipamorelin Preclinical Research
Raun et al. (1998) — Landmark selectivity study in Sprague-Dawley rats (PMID: 9665836). Demonstrated equivalent GH-releasing potency to GHRP-6 and hexarelin, but with 90% less cortisol response and no meaningful ACTH elevation. This established Ipamorelin’s clean hormonal profile.
Hansen et al. (1999) (PMID: 10352464) — Investigated Ipamorelin’s effects on bone mineral density and body composition in aged rats over 12 weeks. Findings included significant increases in bone mineral content, reduced adiposity, and increased lean mass — outcomes attributed to sustained GH/IGF-1 axis activation.
Synergistic Combination Research
Jørgensen et al. (2001) (PMID: 11420165) — Studied co-administration of GHRH analogs with GHSR agonists in porcine models. Co-administration produced 2–4x greater GH AUC compared to either compound in isolation, with the effect being additive to synergistic depending on timing. This provided the pharmacological rationale for the CJC + Ipamorelin combination.
Alba et al. (2006) (PMID: 16648569) — Demonstrated that GHRH analog + GHSR agonist combinations in GH-deficient animal models restored IGF-1 to near-normal levels more effectively than either compound alone, without supraphysiological overshoot. The study highlighted preserved pulsatility as a key benefit of combination vs. exogenous GH therapy.
Comparison: CJC/Ipamorelin vs. Alternative GH Research Stacks
Understanding where CJC-1295 + Ipamorelin fits in the broader landscape of growth hormone research helps researchers select the appropriate tools for their experimental objectives.
CJC-1295 + Ipamorelin vs. CJC-1295 + Tesamorelin (Spartan Strong)
Tesamorelin is an FDA-approved GHRH analog (brand name Egrifta) with a notably strong GH-stimulating profile. In combination studies, pairing CJC-1295 (no-DAC) with Tesamorelin creates a dual-GHRH stimulation protocol — hitting the same receptor pathway with two compounds of different pharmacokinetics. Research applications may include models requiring sustained GHRHR activation with overlapping half-lives.
Our Spartan Strong CJC + Tesamorelin blend is designed for researchers investigating this dual GHRH stimulation approach. In contrast, CJC + Ipamorelin targets two separate receptor families (GHRHR + GHSR) — making it more pharmacologically complementary and theoretically producing greater synergy via independent signaling pathways.
Key comparison summary:
- CJC + Ipamorelin: Dual receptor pathway (GHRHR + GHSR), synergistic GH pulse, preserved pulsatility, minimal off-target hormones
- CJC + Tesamorelin: Single receptor pathway (GHRHR x2), strong GH stimulation, suitable for sustained GHRH-mediated GH research
CJC/Ipamorelin vs. Standalone GH Peptides
Individual GHRH analogs or GH secretagogues produce modest GH elevations compared to the stack. Standalone GHRH analogs typically produce GH increases of 2–5x baseline in research models; standalone GHSR agonists produce comparable results. The CJC + Ipamorelin combination consistently produces 3–8x baseline GH elevation in published models — a meaningful difference for studies requiring robust GH axis stimulation.
Compared to exogenous recombinant GH (rhGH), the combination has a distinct advantage: it stimulates endogenous GH production rather than bypassing it. This preserves physiological feedback mechanisms (IGF-1 negative feedback, somatostatin release) and avoids the supraphysiological IGF-1 spikes associated with rhGH administration — important variables in research design.
Reconstitution and Storage for Research Use
Proper peptide handling is critical for experimental reproducibility. Below are standard laboratory protocols for CJC-1295 and Ipamorelin. For a comprehensive step-by-step guide, see our dedicated article: How to Reconstitute Peptides Safely for R&D.
Reconstitution Protocol
- Solvent: Bacteriostatic water (0.9% benzyl alcohol) is the standard reconstitution medium for both peptides. Sterile water may be used if the solution will be used within 24 hours.
- Volume: Add solvent slowly down the side of the vial — do not inject directly onto the lyophilized powder. Roll gently to mix; never vortex.
- Concentration: Common research concentrations range from 1–2 mg/mL. For the 10 mg blend vial, 5 mL of bacteriostatic water yields 2 mg/mL of combined peptide.
- Sterile filtration: Use a 0.22 µm syringe filter if preparing large volumes or transferring between vessels.
Storage Conditions
- Lyophilized (unreconstituted): Store at -20°C for long-term storage (up to 24 months); 4°C acceptable for up to 3 months if protected from light and moisture.
- Reconstituted solution: Store at 4°C (refrigerator), protected from light. Use within 28–30 days when prepared with bacteriostatic water. Do not freeze reconstituted peptide.
- Temperature sensitivity: Both peptides are sensitive to repeated freeze-thaw cycles, which degrade tertiary structure and reduce biological activity. Aliquot if multiple freeze-thaw cycles are anticipated.
- Light protection: Amber vials or aluminum foil wrap are recommended, as UV exposure accelerates peptide oxidation.
Quality Assessment
Researchers should verify peptide identity and purity via HPLC and mass spectrometry analysis. Spartan Peptides provides third-party CoA documentation with each peptide, including purity (>98%) and molecular weight confirmation. Look for clarity in reconstituted solution — cloudiness, particulates, or color changes indicate degradation.
Frequently Asked Questions
What is the difference between CJC-1295 with DAC and CJC-1295 without DAC in research?
CJC-1295 with DAC (Drug Affinity Complex) binds to serum albumin via a covalent bond, extending its half-life to 6–8 days and producing sustained, non-pulsatile GH elevation. CJC-1295 without DAC (Mod GRF 1-29) has a ~30-minute half-life and produces discrete GH pulses. In combination research with Ipamorelin, the no-DAC form is preferred because it creates physiologically relevant pulsatile GH release rather than a continuous “bleed.”
Why is Ipamorelin considered more selective than other GHRP peptides?
Most GH secretagogues (GHRP-2, GHRP-6, hexarelin) stimulate GHSR but also trigger significant cortisol, prolactin, and ACTH release through off-target receptor interactions. Ipamorelin was specifically engineered for high GHSR selectivity — research shows it produces 90% less cortisol response than GHRP-6 at equivalent GH-releasing doses (Raun et al., 1998). This makes it the preferred GHSR agonist for clean experimental designs.
How does the CJC-1295 + Ipamorelin combination compare to exogenous recombinant GH?
Exogenous recombinant GH (rhGH) bypasses the hypothalamic-pituitary axis entirely, delivering supraphysiological GH directly. This disrupts natural feedback loops — IGF-1 can spike well above physiological ranges and negative feedback is circumvented. CJC-1295 + Ipamorelin stimulates endogenous GH production within the context of physiological regulation: somatostatin feedback remains active, IGF-1 elevation is more proportional, and pulsatile patterns are preserved. For research modeling physiological GH axis function, the peptide combination offers a more naturalistic approach.
What makes CJC-1295 + Ipamorelin research relevant to body composition studies?
Growth hormone has direct lipolytic effects on adipocytes (fat breakdown) and anabolic effects on muscle via IGF-1-dependent and IGF-1-independent pathways. Elevated GH/IGF-1 in research models consistently shows associations with decreased adiposity, increased lean mass, and improved nitrogen retention. The CJC + Ipamorelin combination’s ability to amplify GH pulses while preserving physiological patterns makes it well-suited for studying these body composition mechanisms in preclinical models without the confounding variables introduced by supraphysiological GH doses.
What storage conditions are critical for maintaining CJC-1295 + Ipamorelin stability?
Lyophilized peptide is stable at -20°C for up to 24 months or at 4°C for 3 months. Once reconstituted, solutions should be kept at 4°C and used within 30 days (when using bacteriostatic water). Avoid freeze-thaw cycles on reconstituted solution, protect from UV light, and never heat above room temperature. Signs of degradation include cloudiness, color change, or particulate formation.
Where can I source research-grade CJC-1295 + Ipamorelin for laboratory use?
Research-grade peptides require third-party analytical verification. At Spartan Peptides, our CJC + Ipamorelin 10 mg Blend undergoes independent HPLC and mass spectrometry testing to confirm >98% purity and correct molecular identity. Each lot comes with a Certificate of Analysis. This product is manufactured under strict quality control conditions and supplied exclusively for licensed research use.
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →
⚗️ Research Disclaimer: All content on this page is intended exclusively for licensed researchers, academic institutions, and scientific professionals operating within approved laboratory settings. CJC-1295 and Ipamorelin are research compounds not approved for human clinical use by the FDA or equivalent regulatory authorities. This content does not constitute medical advice, clinical guidance, or a recommendation for use in humans or animals outside of approved research protocols. All research must comply with applicable institutional, local, state, and federal regulations. Spartan Peptides supplies these compounds strictly for in vitro and preclinical research purposes.