Complete Guide to Healing & Recovery Peptides
Written bySpartan Research Team
Healing peptides and recovery compounds represent one of the most prolific areas of preclinical peptide research — a field that has expanded substantially over the past two decades as investigators mapped the molecular machinery governing tissue repair, angiogenesis, and inflammatory resolution. The interest is mechanistically justified: the processes that determine how quickly and completely tissue recovers from injury — wound healing, tendon remodeling, muscle fiber regeneration, gut mucosal repair — are regulated by discrete, targetable signaling pathways, many of which involve endogenous peptides and growth factors. Synthetic research peptides offer a tool for interrogating these pathways with a degree of specificity difficult to achieve with conventional pharmacological agents.
This guide provides a comprehensive, research-grounded overview of the primary recovery peptides currently under scientific investigation. It covers the biology of tissue repair that these compounds target, the distinct mechanisms through which BPC-157, TB-500, and related research compounds interact with healing pathways, and the key preclinical evidence supporting each. It also reviews the research rationale behind combination protocols — specifically the BPC-157/TB-500 combination commonly referenced in the injury recovery literature — and addresses what compound quality standards are required for interpretable research outcomes.
All compounds discussed here are research peptides supplied strictly for laboratory investigation. They are not approved for human consumption and are referenced exclusively in the context of scientific research.
The Biology of Tissue Repair: What Recovery Peptides Target
Tissue healing is not a single event but a coordinated, multi-phase biological process involving overlapping cellular and molecular programs. Understanding this process is a prerequisite to understanding what each class of recovery peptide actually does at the mechanistic level. The repair sequence across most tissue types — skeletal muscle, tendon, ligament, gut mucosa, peripheral nerve — follows a broadly conserved architecture:
Phase 1: Hemostasis and Inflammatory Initiation (Hours 0–72)
Immediately following injury, platelet aggregation and fibrin clot formation establish a provisional matrix. Damaged cells release damage-associated molecular patterns (DAMPs) that activate the innate immune response, recruiting neutrophils and macrophages to the injury site. Macrophages initially adopt a pro-inflammatory (M1) phenotype, secreting TNF-alpha, IL-1beta, IL-6, and reactive oxygen species to clear debris and pathogens. This inflammatory phase is physiologically necessary — its premature suppression impairs downstream repair — but its excessive duration or magnitude is a major driver of fibrotic scarring and incomplete functional recovery.
Phase 2: Proliferative Repair (Days 3–21)
Anti-inflammatory (M2) macrophages predominate by days 3–5, secreting TGF-beta, VEGF, PDGF, and IGF-1 to initiate the proliferative phase. Fibroblasts migrate into the wound and begin synthesizing collagen (initially type III, later remodeled to type I in load-bearing tissues). Angiogenesis — the sprouting of new capillaries from existing vasculature — is driven primarily by VEGF and is critical for delivering oxygen and nutrients to the metabolically active repair zone. Satellite cells (in muscle) and tendon stem cells are activated and begin proliferating to replace lost contractile and structural tissue.
Phase 3: Remodeling (Weeks 3–12+)
The collagen matrix is progressively reorganized, with type III collagen replaced by type I and cross-linking density increasing to restore mechanical strength. Myofibroblasts contract the wound, reducing its area, and eventually undergo apoptosis. In tendons and ligaments, full mechanical property restoration can require 6–12 months even in optimal conditions. The quality of the remodeled matrix — its collagen fiber alignment, cross-link density, and vascular supply — determines the functional outcome and re-injury risk.
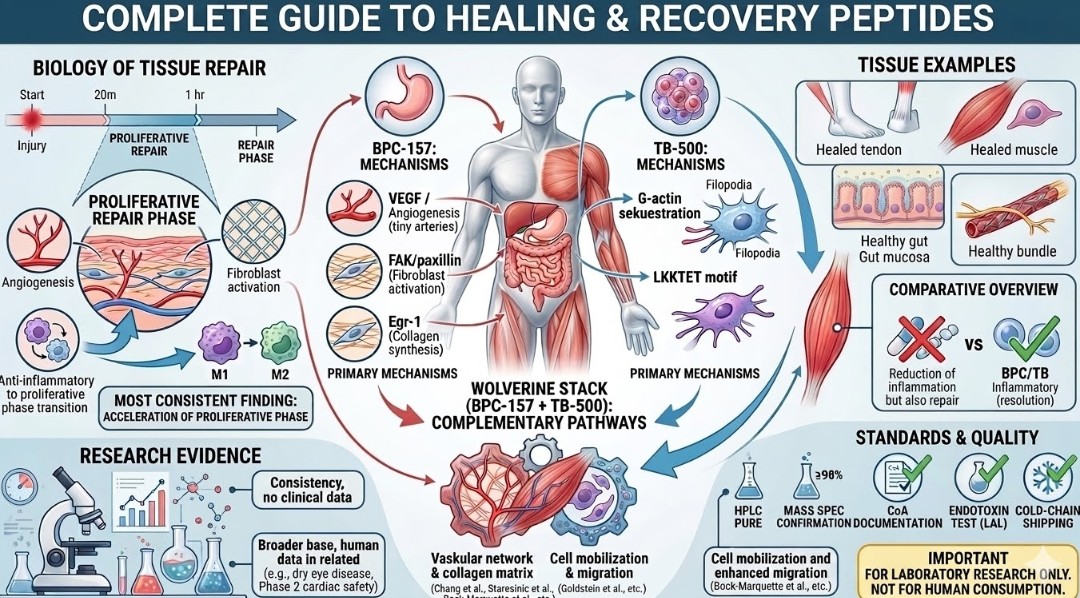
| ★ | Key research context: The most consistent finding across BPC-157 and TB-500 preclinical studies is acceleration of the proliferative phase — earlier angiogenesis, faster fibroblast migration, improved collagen organization — rather than any single mechanism. These compounds appear to amplify the body’s endogenous repair signals rather than introducing an entirely foreign pathway. |
The research peptides discussed in this guide intervene at multiple points in this sequence — most prominently in angiogenesis, fibroblast biology, and the inflammatory-to-proliferative phase transition. Understanding which phase a compound targets is essential for interpreting the preclinical literature correctly.
How Recovery Peptides Work: Primary Mechanism Classes
VEGF Pathway Modulation and Angiogenesis
Vascular endothelial growth factor (VEGF) is the central driver of angiogenesis — the formation of new blood vessels from existing vasculature. In the repair context, angiogenesis is rate-limiting for tissue recovery: without adequate vascular supply, the proliferating fibroblasts and regenerating muscle cells in the repair zone cannot receive sufficient oxygen and nutrients to sustain high-rate protein synthesis. BPC-157 has been documented across multiple preclinical models to upregulate VEGF receptor expression and promote angiogenesis in injured tissue, an effect attributed to activation of the FAK-paxillin signaling pathway in endothelial cells.
Actin Sequestration and Cell Migration (Thymosin Beta-4 Mechanism)
TB-500 (Thymosin Beta-4) operates through a fundamentally different mechanism: it binds G-actin (monomeric actin) via its LKKTET motif, regulating the dynamic balance between filamentous actin (F-actin) and monomeric actin (G-actin) at the leading edge of migrating cells. Actin polymerization dynamics govern the formation of lamellipodia and filopodia — the protrusive structures that drive cell migration. By modulating G-actin availability, TB-500 influences the migration of fibroblasts, endothelial cells, and myoblasts into the injury site, which is a prerequisite for both wound closure and satellite cell-mediated muscle fiber regeneration.
Egr-1 and Collagen Synthesis
Early growth response protein 1 (Egr-1) is a zinc-finger transcription factor that drives expression of multiple extracellular matrix components, including collagen types I and III, fibronectin, and tenascin-C. BPC-157 has been shown in fibroblast cell culture and in vivo tendon models to activate Egr-1 expression, which in turn drives collagen synthesis and matrix organization. This pathway provides a mechanistic explanation for the improved histological collagen organization documented in BPC-157 tendon healing studies beyond what would be expected from angiogenesis alone.
Inflammatory Modulation: Resolution Without Suppression
A critical distinction between the effects of research recovery peptides and classical anti-inflammatory drugs (NSAIDs, corticosteroids) is the relationship to the inflammatory phase. NSAIDs and corticosteroids suppress the inflammatory cascade broadly, which reduces pain but at the cost of impairing downstream proliferative repair — a well-documented phenomenon in tendon and ligament healing research. BPC-157 preclinical data consistently show reduced pro-inflammatory cytokine production (TNF-alpha, IL-6) without complete inflammatory suppression, preserving the physiological signaling necessary for M2 macrophage transition and proliferative phase initiation. This selectivity profile is one of the most mechanistically interesting features of the compound in the repair research literature.
Key Healing and Recovery Peptides: Compound Profiles
- BPC-157 (Body Protection Compound 157)
BPC-157 is a synthetic pentadecapeptide of 15 amino acids (Ala-Gly-Glu-Gly-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly) derived from a sequence within the gastric juice protein BPC. It was first characterized by Sikiric et al. at the University of Zagreb, whose laboratory has produced the majority of the foundational BPC-157 preclinical literature. The compound has been investigated across an unusually broad range of injury models — tendon, ligament, muscle, bone, gut mucosa, peripheral nerve, and skin — consistently demonstrating accelerated healing relative to control conditions.
The mechanistic breadth of BPC-157 is unusual for a single compound. Established preclinical findings include: upregulation of VEGF receptor expression and promotion of angiogenesis; activation of FAK-paxillin signaling in endothelial cells and fibroblasts; induction of Egr-1 transcription and downstream collagen synthesis; modulation of nitric oxide (NO) system activity with organ-protective effects; and selective attenuation of pro-inflammatory cytokines without complete inflammatory suppression. No single receptor has been identified as the primary BPC-157 target, which distinguishes it mechanistically from classical receptor agonists and has made comprehensive mechanistic characterization an ongoing research challenge.
Body of evidence highlights:
- Staresinic et al. (2003): Accelerated Achilles tendon healing in rats with improved histological collagen organization and biomechanical properties vs. control.
- Chang et al. (2011): Accelerated healing of transected quadriceps tendon; BPC-157 promoted tendon outgrowth and cell survival in both in vivo and in vitro models.
- Sikiric et al. (2018): Comprehensive review of BPC-157’s systemic organ-protective effects across GI, cardiovascular, and musculoskeletal injury models, emphasizing the NO system modulation as a unifying mechanistic thread.
- Gut mucosa models: BPC-157 has demonstrated consistent cytoprotective activity in gastric and intestinal injury models, accelerating ulcer healing and protecting against NSAID-induced mucosal damage — a finding with implications for the gut-systemic recovery axis.
For the specific joint and tendon repair literature, see Joint and Tendon Repair. For a research-framed review of what the side effect data show, see BPC-157 Side Effects Research.
- TB-500 (Thymosin Beta-4)
TB-500, the research compound form of Thymosin Beta-4, is a 43-amino acid peptide that is among the most abundant intracellular peptides in mammalian cells. Endogenous Thymosin Beta-4 is constitutively expressed in platelets, neutrophils, and macrophages — all key cellular actors in the early repair phase — and is released in elevated concentrations at injury sites. Its exogenous administration in research models represents an amplification of an endogenous wound healing signal rather than the introduction of a foreign mechanism.
The LKKTET actin-binding motif is the functional core of Thymosin Beta-4’s biological activity. By sequestering G-actin, it influences the protrusive dynamics of cell migration across multiple cell types relevant to repair: endothelial cells (angiogenesis), fibroblasts (matrix deposition), and myoblasts (muscle fiber regeneration). Beyond actin biology, TB-500 has been shown to upregulate the expression of laminin-5, an extracellular matrix component critical for epithelial cell migration, and to promote cardiomyocyte survival following ischemic injury in animal models.
Key preclinical findings:
- Malinda et al. (1999): TB-500 promoted dermal wound healing in rats, accelerating wound closure and increasing angiogenesis relative to saline controls.
- Goldstein et al. (2012): Review of cardiac and skeletal muscle repair data showed consistent findings of stem cell recruitment, angiogenesis promotion, and anti-fibrotic signaling across injury models.
- Bock-Marquette et al. (2004): TB-500 demonstrated cardioprotective effects in murine myocardial infarction models, promoting cardiomyocyte survival and neovascularization — establishing the peptide’s activity in non-musculoskeletal repair contexts.
- Anti-fibrotic data: Multiple models have shown TB-500 reduces collagen deposition in a disorganized, fibrotic pattern relative to controls, suggesting it promotes organized matrix remodeling over scarring.
For a mechanistic comparison of the two primary thymosin family members, see Thymosin Beta-4 vs. Alpha-1. For the acceleration of recovery data specifically, see Accelerating Recovery and Repair.
- Wolverine Stack: BPC-157 + TB-500
The Wolverine research compound combines BPC-157 and TB-500 within a single formulation, targeting the mechanistic complementarity between these two peptides in tissue repair biology. The research rationale is straightforward: BPC-157 drives angiogenesis, fibroblast activation, and collagen organization through VEGF/FAK/Egr-1 pathways, while TB-500 facilitates cell migration and satellite cell recruitment through actin sequestration and laminin-5 upregulation. These two mechanisms address different rate-limiting steps in the repair sequence — vascular supply and matrix architecture on one hand, cellular mobilization and migration on the other — making them mechanistically non-redundant.
Preclinical studies examining each compound individually have consistently demonstrated their respective mechanistic profiles across overlapping injury models. Controlled studies examining their co-administration are less numerous in the peer-reviewed literature — partly because the pharmaceutical development rationale for a combination product requires a different study design than single-compound mechanistic research — but the physiological logic of combining complementary repair mechanisms is consistent with established principles of wound healing biology.
The combination also reflects a broader principle in repair biology: that tissue healing is multi-factorial, and interventions targeting single pathway nodes produce smaller effects than those addressing multiple rate-limiting steps simultaneously. In research models where both vascular insufficiency and impaired cell migration contribute to delayed repair, a combination addressing both constraints would be expected to outperform either compound individually.
Spartan Peptides offers the Wolverine BPC-157 + TB-500 research compound at ≥98% HPLC purity with full CoA documentation. For an overview, see the science behind this combination
Comparative Overview: Healing & Recovery Peptides at a Glance
| Compound | Primary Mechanism | Key Preclinical Finding | Tissue Targets | Compound Class |
|---|---|---|---|---|
| BPC-157 | VEGF/FAK/Egr-1 — angiogenesis, fibroblast activation, collagen synthesis | Accelerated tendon-to-bone healing; improved collagen histology; gut mucosal cytoprotection | Tendon, ligament, muscle, gut mucosa, nerve | Gastric peptide fragment |
| TB-500 | G-actin sequestration (LKKTET) — cell migration, satellite cell recruitment, anti-fibrosis | Accelerated wound closure; stem cell recruitment; cardioprotection in MI models | Skin, muscle, cardiac, systemic | Thymosin-derived peptide |
| Wolverine (BPC-157 + TB-500) | Complementary: VEGF/ECM pathways + actin/migration pathways | Addresses both the vascular supply and the cellular mobilization rate-limiting steps simultaneously | Musculoskeletal, connective tissue, systemic | Combination stack |
Tissue-Specific Research: What the Data Show by Injury Type
Tendon and Ligament Repair
Tendons and ligaments are among the most thoroughly studied tissues in the BPC-157 and TB-500 literature, reflecting the high prevalence of tendon injuries in athletic and clinical populations and the notoriously slow healing capacity of these relatively avascular structures. The key constraints on tendon healing — limited intrinsic vascularity, slow fibroblast migration, and the mechanical demands placed on tissue during the repair phase — map directly onto the known mechanistic effects of both compounds.
BPC-157 tendon data from Staresinic et al. (2003) and Chang et al. (2011) showed not only faster healing timelines but improved biomechanical properties at matched timepoints — a distinction from studies reporting only timeline acceleration, as it suggests qualitatively better tissue rather than simply faster scarring. The gut health dimension of BPC-157 research is also increasingly recognized as relevant to systemic recovery: gut mucosal integrity influences nutrient absorption, inflammatory tone, and systemic immune regulation.
Skeletal Muscle Repair
Skeletal muscle has considerably greater intrinsic healing capacity than tendon due to its superior vascular supply and the presence of satellite cells — resident muscle stem cells that activate upon injury to proliferate and fuse with damaged fibers. Nevertheless, severe or repeated muscle injuries can lead to fibrotic replacement of contractile tissue, with lasting functional deficits. TB-500’s role in satellite cell migration and its anti-fibrotic signaling profile make it a compound of particular interest in muscle injury models.
Preclinical data from Malinda et al. and subsequent muscle injury models show TB-500 accelerating the recruitment of myoblasts to injury sites and reducing the extent of fibrotic collagen deposition. BPC-157’s angiogenic effects complement this by ensuring adequate perfusion of the regenerating fiber bed.
Gut Mucosal Repair
BPC-157’s gastric origin — derived from a sequence within human gastric juice protein — correlates with its particularly robust activity in gastrointestinal injury models. Preclinical studies have documented BPC-157 accelerating healing of gastric ulcers, protecting intestinal mucosa from NSAID-induced damage, and attenuating colitis-like inflammation in rodent models. The mechanistic basis involves cytoprotective NO pathway modulation and direct fibroblast-stimulating activity in the lamina propria.
The gut-repair dimension of BPC-157 research is also relevant to systemic recovery biology. Gut mucosal barrier integrity influences systemic inflammatory tone through regulation of bacterial translocation and endotoxin passage. Research in this area positions BPC-157 as potentially relevant to recovery biology beyond direct musculoskeletal injury contexts.
Nerve and Vascular Repair
Peripheral nerve and vascular repair represent emerging areas of BPC-157 research, with preclinical data documenting accelerated nerve fiber regeneration and improved functional recovery following crush and transection injuries. The angiogenic activity of BPC-157 is mechanistically relevant here — nerve regeneration requires axon sprouting along a vascular scaffold, and inadequate angiogenesis is a rate-limiting factor in peripheral nerve recovery. Several Sikiric group papers have documented BPC-157’s effects on peripheral nerve regeneration in rat sciatic nerve models, showing improved axon myelination and functional recovery metrics relative to controls.
Recovery Peptides vs. Conventional Anti-Inflammatory Agents: A Research Perspective
A recurring theme in the healing peptide literature is the mechanistic contrast between research recovery compounds and the standard pharmacological approach to injury management — non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids. This comparison is important for understanding why the preclinical evidence for peptides like BPC-157 has attracted research interest even in contexts where established anti-inflammatory drugs are available.
NSAIDs inhibit cyclooxygenase (COX) enzymes, reducing prostaglandin synthesis and thereby attenuating the inflammatory phase of repair. This produces effective short-term pain reduction but has a documented negative effect on the proliferative repair phase: prostaglandins are required for fibroblast activation, VEGF expression, and collagen synthesis, and their suppression delays functional tissue recovery in tendon and ligament models (Warden, 2010; Jones et al., 2021). Corticosteroids produce broader suppression of the inflammatory cascade, with even more pronounced negative effects on collagen synthesis and long-term mechanical properties of healed tissue.
BPC-157’s inflammatory profile — selectively attenuating pro-inflammatory cytokines while preserving the VEGF and proliferative signals necessary for repair — represents a mechanistically distinct approach. It targets the resolution of the inflammatory phase rather than its suppression, which in preclinical models is associated with faster transition to the proliferative phase without the collagen synthesis impairment documented with NSAIDs. This mechanistic distinction is among the most scientifically interesting aspects of BPC-157 research and continues to drive investigation into its practical applications.
Research Evidence: Synthesis of the Preclinical Literature
Consistency and Breadth of BPC-157 Data
The BPC-157 literature is notable for its breadth across injury types and its consistency of directional findings. Across more than 50 published preclinical studies — predominantly from the Sikiric group at Zagreb but increasingly from independent laboratories — BPC-157 has demonstrated accelerated healing relative to control conditions in every tissue type examined. This consistency across mechanistically diverse injury models (tendon transection, muscle crush, gastric ulcer, colon anastomosis, sciatic nerve crush, bone fracture) is unusual in the preclinical peptide literature and has contributed to sustained research interest despite the absence of controlled human clinical trial data.
The primary limitation of the BPC-157 evidence base is its near-exclusive preclinical character. No Phase 1 or Phase 2 human clinical trials have been published, meaning the translation of animal model findings to human biology remains unvalidated. This is a standard caveat for research compounds without clinical development programs, and it does not diminish the mechanistic value of the preclinical data for research purposes.
TB-500 Human and Clinical Adjacency Data
The Thymosin Beta-4 evidence base includes a broader range of study types than BPC-157, partly because thymosin peptides have undergone clinical investigation in contexts adjacent to musculoskeletal repair. RegeneRx Biopharmaceuticals conducted Phase 2 trials of Thymosin Beta-4 in dry eye disease and neurotrophic corneal ulcer, providing human tolerability and pharmacokinetic data. The compound demonstrated a favorable safety profile in these contexts, which provides a supportive context for the broader preclinical body of work, even though these indications are distinct from the musculoskeletal applications most studied preclinically.
Phase 1 cardiac safety data for Thymosin Beta-4 (from the AMPERE study) documented tolerability in acute MI patients, providing additional human pharmacokinetic and safety context. These human data do not directly validate the musculoskeletal repair applications but establish the compound’s safety profile in human subjects.
What the Data Do and Do Not Show
A rigorous reading of the recovery peptide literature requires distinguishing what the data demonstrate from what they suggest. The preclinical data for BPC-157 and TB-500 consistently demonstrate: (1) acceleration of healing timelines relative to untreated controls; (2) improved histological organization of repaired tissue; (3) enhanced angiogenesis in the repair zone; (4) reduced fibrotic scarring in some models. What the data do not demonstrate — because the studies have not been conducted — is: (1) controlled human clinical efficacy in defined injury populations; (2) optimal administration parameters for human research; (3) long-term outcome data beyond the repair phase. These are not criticisms of the science; they are accurate characterizations of where the evidence base currently stands.
| ★ | Research note: The absence of human clinical trial data for BPC-157 is primarily a reflection of the research compound’s status rather than a safety concern. The compound has not undergone formal pharmaceutical development, meaning Phase 1 trials — which are the prerequisite for any clinical data — have not been conducted. |
Research-Grade Quality Requirements for Recovery Peptides
Tissue repair research is a field where compound quality has an outsized effect on result interpretability. BPC-157 and TB-500 both act through pathways that are sensitive to structural integrity: the LKKTET actin-binding motif of TB-500 requires a correct amino acid sequence and conformation to bind G-actin; BPC-157’s multi-pathway activity has been characterized for the specific 15-amino-acid sequence, and truncation or oxidation artifacts would produce a compound with different and unpredictable biological activity.
The critical quality parameters for repair peptide research are:
- HPLC purity ≥98%: The primary analytical criterion. Confirms that the major chromatographic peak — corresponding to the target compound — accounts for ≥98% of UV-absorbing material at 214–220nm.
- Mass spectrometry confirmation: ESI-MS or MALDI-TOF verifying molecular weight matches the theoretical value for the target sequence. For BPC-157 (MW ~1419 Da) and TB-500 (MW ~4963 Da), mass confirmation rules out common synthesis errors, including deletion sequences and oxidized methionine variants.
- Certificate of Analysis (CoA): Batch-specific documentation of purity data, mass confirmation, and synthesis date. Required for research traceability and protocol reproducibility.
- Endotoxin testing: Limulus amebocyte lysate (LAL) testing for bacterial endotoxin is essential for in vivo repair research. Lipopolysaccharide contamination produces a systemic inflammatory response that would directly confound the inflammatory phase measurements central to repair research.
- US-origin cold-chain shipping: Peptides are thermolabile, and BPC-157 in particular is susceptible to degradation under ambient temperature conditions. Domestic sourcing with documented cold-chain management materially reduces the risk of receiving a degraded compound that will not reproduce expected receptor-level activity.
Frequently Asked Questions
Q: What makes BPC-157 mechanistically different from other healing peptides?
BPC-157 is unusual in the peptide healing research landscape for two reasons: its mechanistic breadth and the consistency of positive findings across an unusually wide range of tissue types. Most healing-relevant compounds target a single pathway — a specific growth factor receptor, a cytokine, or a matrix metalloproteinase. BPC-157 has documented activity across VEGF/angiogenesis pathways, FAK-paxillin fibroblast signaling, Egr-1-driven collagen synthesis, and NO system modulation. No single primary receptor has been identified, which makes comprehensive mechanistic characterization challenging but also suggests the compound’s effects are not easily abolished by the loss or downregulation of any single pathway component. This multi-target profile is consistent with its origin as a fragment of a gastric cytoprotective protein, which would be expected to have evolved broad tissue-protective rather than single-mechanism activity.
Q: How does TB-500 complement BPC-157 in combination research protocols?
BPC-157 and TB-500 address different rate-limiting steps in the tissue repair sequence. BPC-157’s primary contributions are to the vascular and matrix architecture of repair: it drives angiogenesis through VEGF pathways, activates fibroblast collagen synthesis through Egr-1, and modulates the inflammatory-to-proliferative phase transition. TB-500’s primary contribution is to cellular mobilization: by sequestering G-actin and regulating cell migration dynamics, it facilitates the movement of fibroblasts, endothelial cells, and myoblasts into the injury zone. In research models where both vascular supply and cellular recruitment are limiting factors, combining compounds that address each constraint independently is expected to produce additive effects, which is the biological logic underlying the Wolverine combination protocol.
Q: Why is gut mucosal repair relevant to systemic recovery research?
The gut mucosa is a critical interface between the external environment and the systemic circulation. Mucosal barrier integrity prevents translocation of bacterial endotoxin (lipopolysaccharide) from the intestinal lumen into the bloodstream — a process that, when it occurs, produces systemic inflammatory signaling that elevates circulating IL-6, TNF-alpha, and CRP. In the context of musculoskeletal injury research, systemic inflammatory tone is a determinant of the magnitude and duration of the inflammatory phase at the injury site. BPC-157’s documented gut cytoprotective effects, therefore, have theoretical systemic relevance beyond the direct gut injury context: a compound that maintains mucosal barrier integrity may also attenuate the systemic inflammatory load that slows peripheral tissue repair.
Q: What is the current status of human clinical data for BPC-157 and TB-500?
BPC-157 has no published human clinical trial data. It has not undergone formal pharmaceutical development, and therefore, no Phase 1, Phase 2, or Phase 3 human studies exist in the peer-reviewed literature. All efficacy data are from preclinical animal models. TB-500 (Thymosin Beta-4) has more advanced human data: Phase 2 trials in dry eye disease and neurotrophic corneal ulcer have been completed and published, providing human tolerability and pharmacokinetic information. Phase 1 cardiac safety data also exist from the AMPERE study. However, controlled clinical trials examining the musculoskeletal repair applications most studied preclinically have not been conducted. For both compounds, the research use case is therefore grounded in a substantial but exclusively preclinical evidence base.
Q: What quality standards should research protocols specify for BPC-157 and TB-500?
Research protocols should specify: HPLC-verified purity ≥98% with UV detection at 214–220nm; mass spectrometry confirmation (ESI-MS or MALDI-TOF) of correct molecular weight for the target sequence; batch-specific Certificate of Analysis documentation; and, for in vivo models, LAL endotoxin testing with defined acceptable limits (typically <1 EU/mg). Cold-chain management from synthesis to delivery is a critical but frequently underspecified parameter: ambient temperature exposure during shipping degrades peptide integrity in ways that are not always detectable without repeat analytical testing. Protocols should require documentation of cold-chain maintenance or conduct incoming analytical testing before use.
Conclusion
The healing and recovery peptide research field is distinguished by its mechanistic depth and the breadth of tissue systems under investigation. BPC-157 has generated the most extensive single-compound preclinical evidence base in this category, documenting consistent pro-healing effects across tendon, ligament, muscle, gut mucosa, and nerve tissue through a multi-pathway mechanism that sets it apart from classical single-target receptor agonists. TB-500 complements this profile through its actin sequestration mechanism, targeting cell migration and satellite cell recruitment — processes that are mechanistically distinct from but biologically synergistic with the angiogenic and matrix-organizing effects of BPC-157. Together, as the Wolverine combination, they represent a research tool designed around the multi-factorial nature of tissue repair biology.
For researchers working in tissue repair and recovery biology, Spartan Peptides maintains a dedicated repair and recovery research catalog covering BPC-157, TB-500, and the Wolverine combination compound — all supplied at ≥98% HPLC-verified purity with full Certificate of Analysis documentation. For a broader view of the peptide research landscape, see What Are Peptides?.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
Jones, A. et al. (2021). The effect of non-steroidal anti-inflammatory drugs on tendon healing: a systematic review and meta-analysis. Journal of Orthopaedic Research, 39(4), 741–751.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →