Complete Guide to Sexual Health Peptides
Written bySpartan Research Team
Sexual health peptides represent a mechanistically specific category within reproductive and neuroendocrine research — one where the relevant biology is well-characterized and where peptide compounds offer tools for studying receptor systems that conventional pharmacological agents cannot access with equivalent precision. The question driving this research is straightforward: which signaling pathways govern sexual arousal, gonadotropin release, and reproductive axis function, and can targeted research compounds modulate these pathways in controlled laboratory models?
This guide covers the primary sexual health and libido peptides currently under scientific investigation. PT-141 (bremelanotide) and Kisspeptin address two distinct levels of the reproductive neuroendocrine hierarchy: PT-141 acts centrally at hypothalamic and limbic melanocortin receptors to modulate sexual arousal circuitry directly, while Kisspeptin acts at GnRH neurons to regulate the entire pulsatile gonadotropin axis that governs gonadal function. Together, they represent both ends of the sexual health research spectrum — the acute, centrally-mediated arousal response and the chronic, axis-level hormonal regulation that determines baseline reproductive capacity.
All compounds discussed here are research peptides supplied strictly for laboratory investigation. They are not approved for human consumption and are referenced exclusively in the context of scientific research.
The Neuroendocrinology of Sexual Function: What Sexual Health Peptides Target
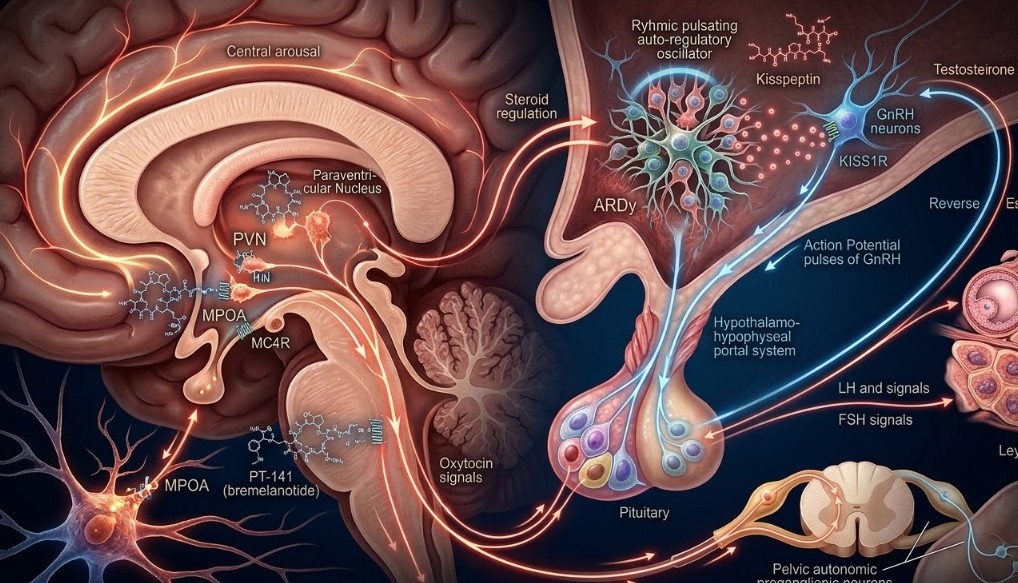
Sexual function is regulated at multiple anatomical levels simultaneously — the central nervous system (hypothalamus, limbic system, brainstem), the pituitary, the gonads, and peripheral vascular and neuromuscular systems. Research peptides in the sexual health category primarily target the CNS and hypothalamic-pituitary-gonadal (HPG) axis, where the initiating signals for both arousal and gonadal steroid production originate.
The Melanocortin System and Central Arousal
The melanocortin system comprises a family of peptide receptors (MC1R through MC5R) activated by melanocyte-stimulating hormone (MSH) peptides and ACTH, all derived from the precursor protein proopiomelanocortin (POMC). In the context of sexual function, MC3R and MC4R in the hypothalamus and limbic system are the most relevant receptors. MC4R in particular has been shown to modulate sexual arousal behavior in animal models: MC4R knockout mice display reduced sexual behavior, and selective MC4R agonism with synthetic peptides produces increased mounting behavior and erection in male rodents, and increased lordosis behavior in female rodents.
The hypothalamic paraventricular nucleus (PVN) contains a high density of MC4R-expressing neurons, and melanocortin signaling in the PVN activates oxytocin-releasing neurons that project to spinal cord autonomic centers governing genital vascular response and arousal. This hypothalamic → spinal cord pathway — mediated by oxytocin release from PVN neurons following MC4R activation — is the proposed central mechanism by which melanocortin agonists produce their pro-erectile and pro-arousal effects in animal models.
The HPG Axis: GnRH Pulses and Gonadotropin Release
The hypothalamic-pituitary-gonadal axis operates through pulsatile signaling that is architecturally different from most other endocrine axes. Gonadotropin-releasing hormone (GnRH) is released from hypothalamic neurons in discrete pulses at approximately 60–90-minute intervals in adult males and at varying frequencies across the menstrual cycle in females. These GnRH pulses drive pulsatile LH and FSH secretion from anterior pituitary gonadotroph cells — and this pulsatility is not incidental. Continuous GnRH stimulation (as with long-acting GnRH agonist drugs used in prostate cancer) paradoxically desensitizes pituitary GnRH receptors and suppresses LH/FSH. Only pulsatile GnRH release produces tonic gonadotropin secretion.
LH drives gonadal steroid synthesis: testosterone in Leydig cells (male), estradiol and progesterone in granulosa/luteal cells (female). FSH drives gametogenesis: spermatogenesis in Sertoli cells, follicular development in granulosa cells. The gonadal steroids provide negative feedback to the hypothalamus and pituitary to regulate GnRH/LH/FSH secretion through sex steroid receptor signaling at both levels.
Kisspeptin Neurons as the Master Regulator of GnRH Release
Kisspeptin neurons in the arcuate nucleus (ARC) and anteroventral periventricular nucleus (AVPV) represent the primary upstream regulators of GnRH pulse generation. Kisspeptin (the peptide) is the endogenous ligand for the KISS1R (formerly GPR54) receptor on GnRH neurons, and kisspeptin binding produces the calcium-mediated depolarization of GnRH neurons that triggers GnRH pulse release. Kisspeptin signaling is therefore the proximate stimulus for GnRH secretion — and by extension, for the entire downstream HPG axis function.
The discovery of kisspeptin’s role in GnRH regulation came from human genetics: loss-of-function mutations in KISS1R were found to cause hypogonadotropic hypogonadism in humans — a failure of pubertal development and gonadal function. This established kisspeptin/KISS1R as a non-redundant, genetically validated regulator of the HPG axis — the kind of evidence base that is rare in peptide neuroendocrinology and that has driven substantial subsequent research into kisspeptin as a research tool for studying HPG axis biology.
| ★ | Research context: PT-141 and Kisspeptin act at completely different levels of the reproductive neuroendocrine hierarchy and produce different experimental endpoints. PT-141 studies use behavioral, vascular, and functional arousal measures as primary endpoints; Kisspeptin studies use LH pulse frequency and amplitude, FSH levels, testosterone or estradiol concentrations, and follicular development as primary endpoints. Combining data from both research areas requires attention to these different outcome frameworks. |
Sexual Health Research Peptides: Compound Profiles
- PT-141 (Bremelanotide)
PT-141, also known as bremelanotide, is a cyclic heptapeptide melanocortin receptor agonist developed from PT-14, which was itself derived from Melanotan II — a synthetic analogue of alpha-MSH originally studied for tanning effects.
The clinical development of bremelanotide has been the most advanced of any compound in the sexual health peptide category: the FDA approved an intranasal formulation (Vyleesi) for hypoactive sexual desire disorder (HSDD) in premenopausal women in 2019 — making bremelanotide/PT-141 one of the very few research peptides that has completed the pharmaceutical development pathway to FDA approval. The research data underlying this approval provide a relatively well-characterized clinical evidence base. A detailed research overview is available in our PT-141 Bremelanotide: Enhancing Sexual Wellness Research.
PT-141 Mechanism: MC3R/MC4R Agonism and CNS Arousal
PT-141’s mechanism centers on agonism at MC3R and MC4R in hypothalamic and limbic brain regions. Unlike PDE5 inhibitors (sildenafil, tadalafil), which act peripherally by increasing penile vascular blood flow through cGMP accumulation, PT-141 acts centrally — its pro-arousal effects are mediated by CNS receptor activation rather than peripheral vascular changes. This mechanistic distinction is research-relevant because central and peripheral arousal pathways are experimentally separable, and PT-141 provides a tool for studying the CNS contribution to sexual arousal independently of the peripheral vascular component.
The downstream pathway from MC4R activation to genital arousal response involves several sequential steps: MC4R activation in the PVN → oxytocin release from PVN neurons → oxytocin action on spinal cord autonomic preganglionic neurons → parasympathetic activation of pelvic vasculature → genital vascular engorgement and arousal response. This pathway was characterized in part through experiments with oxytocin receptor antagonists, which blunted the pro-erectile effects of MC4R agonism in rat models — providing pharmacological evidence for the oxytocin-dependent component of the mechanism.
MC3R activation in limbic structures — particularly the medial preoptic area (MPOA) and nucleus accumbens — contributes to the motivational and rewarding dimensions of sexual behavior. The MPOA is one of the most studied brain regions for sexual motivation, and MC3R signaling there activates dopaminergic reward circuits that modulate the appetitive (wanting/seeking) component of sexual behavior, distinct from the consummatory (performance) component. PT-141’s activity at both receptor subtypes in these two anatomical regions — the PVN for the autonomic/vascular component and the limbic system for the motivational component — is consistent with its documented effects on both arousal and sexual motivation in research models.
PT-141 Clinical Evidence
The pharmaceutical development program for bremelanotide produced controlled human trial data that is unusually robust relative to most research peptides. Phase 2 and Phase 3 randomized, placebo-controlled trials in premenopausal women with HSDD — enrolling hundreds of subjects with primary endpoints of satisfying sexual events (SSEs) and sexual desire scores — demonstrated statistically significant improvements over placebo on both primary endpoints. The effect sizes were moderate: approximately 0.5–1 additional SSE per 4 weeks above placebo, and a 1.2–1.5 point improvement on a sexual desire score scale, consistent with an active agent in this indication.
The approved formulation uses an intranasal route in a one-time-use autoinjector format, with activity onset at 45–60 minutes and duration of approximately 8–12 hours. The primary safety findings from the trials included transient nausea (the most common adverse event, occurring in approximately 40% of subjects) and transient blood pressure increases that resolved within 12 hours. Subjects with cardiovascular disease were excluded from the trials.
In male research models, the mechanism is identical, but the experimental endpoints differ. Penile erection studies in rats and the small male human trial data documented dose-dependent erection responses following PT-141 administration, consistent with the PVN → oxytocin → spinal cord mechanism. The female data are more extensively characterized because HSDD was the intended pharmaceutical indication, but the mechanistic evidence for both sexes is grounded in the same MC4R/MC3R receptor pharmacology.
For a research-framed overview of PT-141 data in female sexual health models, see our PT-141 for Women: Libido Research Overview. Spartan Peptides supplies PT-141 research peptide at ≥98% HPLC purity with full CoA documentation.
- Kisspeptin
Kisspeptin is a family of peptides (Kp-10, Kp-13, Kp-14, Kp-54) derived from the KISS1 gene product through proteolytic processing — all sharing a C-terminal decapeptide sequence (the RF-amide motif) that is required for KISS1R binding and receptor activation. The naming reflects the discovery context: KISS1 was originally identified as a metastasis-suppressor gene in melanoma (hence the ‘KISS’ prefix, referring to Hershey, PA, the location of the cancer research lab), and its role in reproductive biology was discovered later through the human genetics of hypogonadotropic hypogonadism. Full kisspeptin biology and research data are reviewed in our Kisspeptin: Fertility Peptide Explained.
Kisspeptin Mechanism: KISS1R Activation and GnRH Pulse Generation
Kisspeptin binds KISS1R — a Gq-coupled GPCR — on GnRH neurons in the hypothalamus. Gq coupling activates phospholipase C, generating IP3 and DAG, which elevate intracellular calcium and activate protein kinase C. The calcium elevation depolarizes the GnRH neuron, triggering an action potential and GnRH pulse release into the hypothalamo-hypophyseal portal system. The downstream effects — LH and FSH pulsatile secretion from the anterior pituitary, followed by gonadal steroid synthesis and gametogenesis — follow from GnRH pulse amplitude and frequency in the manner already described.
The ARC kisspeptin neurons co-express neurokinin B (NKB) and dynorphin A alongside kisspeptin, forming the KNDy neuron population (Kisspeptin/Neurokinin B/Dynorphin). NKB acts at NK3 receptors on neighboring KNDy neurons to propagate kisspeptin pulse initiation, while dynorphin acts at kappa-opioid receptors on the same cells to terminate the pulse, creating an auto-regulatory oscillator within the ARC that generates the 60–90 minute GnRH pulse rhythm. Kisspeptin exogenous administration experiments have been used extensively to characterize this oscillator: an acute kisspeptin bolus reliably triggers an LH pulse in research subjects across species, providing a pharmacological probe for ARC oscillator sensitivity.
The AVPV kisspeptin population has a different function: in female rodents, AVPV kisspeptin neurons are responsible for the preovulatory LH surge — the large-amplitude LH release that triggers ovulation — and their activation is dependent on the positive feedback of rising estradiol during the follicular phase. This population is sexually dimorphic: female rodents have more AVPV kisspeptin neurons than males, consistent with their role in the cyclic LH surge that occurs only in females. This dimorphism and its functional consequences make AVPV kisspeptin biology a research area of direct relevance to reproductive physiology and fertility research.
Kisspeptin Human Clinical Data
Kisspeptin has been studied in human clinical research to an unusual extent for a research peptide, primarily through the work of Waljit Dhillo’s group at Imperial College London. Intravenous kisspeptin administration in healthy male volunteers produced dose-dependent LH pulse triggering with minimal side effects (Dhillo et al., 2005, Journal of Clinical Endocrinology & Metabolism). Subsequent studies characterized kisspeptin pharmacokinetics, LH response dose-response relationships, and the effects of exogenous sex steroid modulation on kisspeptin sensitivity.
In women with functional hypothalamic amenorrhea — a condition characterized by HPG axis suppression due to energy deficit or stress — kisspeptin administration successfully restored pulsatile LH secretion in a controlled trial (Jayasena et al., 2009), demonstrating that the underlying GnRH neurons retained functional competence even when endogenous kisspeptin drive was insufficient. This finding established kisspeptin as a potential research tool for studying HPG axis suppression and recovery, with translational relevance to conditions of hypothalamic hypogonadism.
Kisspeptin has also been studied as a trigger for ovarian hyperstimulation in IVF protocols, as an alternative to hCG for final oocyte maturation. Phase 2 data from Jayasena et al. (2014) showed comparable oocyte yields with a potentially lower risk of ovarian hyperstimulation syndrome (OHSS) relative to standard hCG triggers, reflecting kisspeptin’s physiological mechanism — triggering endogenous LH surge through GnRH rather than directly mimicking LH with a pharmacological dose. The IVF trigger data represent the most applied clinical use of kisspeptin in the published literature.
Spartan Peptides supplies Kisspeptin research compound at ≥98% HPLC purity. For an overview of kisspeptin’s reproductive hormone biology, see Regulating Hormones and Reproductive Health with Kisspeptin.
Comparative Overview: Sexual Health Research Peptides
| Compound | Primary Mechanism | Key Research Finding | Research Endpoint | Class |
|---|---|---|---|---|
| PT-141 (Bremelanotide) | MC3R/MC4R agonism in hypothalamus and limbic system; oxytocin-mediated spinal cord activation | FDA-approved (Vyleesi) in HSDD; Phase 3 RCT demonstrated SSE improvement vs. placebo in premenopausal women | Sexual arousal, desire scores, satisfying sexual events (SSEs) | Cyclic heptapeptide; alpha-MSH analogue |
| Kisspeptin | KISS1R (Gα-GPCR) agonism on GnRH neurons; GnRH pulse triggering; HPG axis activation | Dose-dependent LH pulse triggering in human volunteers (Dhillo et al., 2005); restored pulsatile LH in hypothalamic amenorrhea (Jayesena et al., 2009) | LH/FSH pulsatility, gonadal steroid levels, and ovulation induction | Neuropeptide; RF-amide family |
PT-141 and PDE5 Inhibitors: Mechanistic Differences in Sexual Health Research

A frequent comparison in sexual health research is between PT-141/bremelanotide (a central, melanocortin-mediated mechanism) and PDE5 inhibitors such as sildenafil and tadalafil (a peripheral, vascular mechanism). These comparisons are research-relevant because the two compound classes target different anatomical components of the sexual response and therefore provide different information about which physiological factors are rate-limiting in a given research model.
PDE5 inhibitors work by blocking the phosphodiesterase enzyme that degrades cyclic GMP (cGMP) in smooth muscle cells of the corpus cavernosum (and in female genital vascular tissue). Elevated cGMP relaxes smooth muscle, increasing blood flow to the erectile tissue. This mechanism requires prior nitric oxide (NO) synthesis — triggered by sexual stimulation, activating non-adrenergic, non-cholinergic (NANC) neurons — because NO is the upstream activator of guanylate cyclase that generates cGMP in the first place. PDE5 inhibitors, therefore, amplify an ongoing vascular response but do not initiate it in the absence of prior sexual stimulation.
PT-141’s central mechanism is independent of this peripheral vascular pathway. By activating MC4R in the PVN to release oxytocin, it initiates the spinal cord autonomic activation that drives genital vascular response even without requiring prior nitric oxide stimulus from peripheral NANC neurons. In animal models of arousal disorders where PDE5 inhibitor effects are attenuated (because NO synthesis is impaired), PT-141 retains activity through its central mechanism — a finding that has positioned it as a research tool for studying arousal biology in models where peripheral vascular changes are not the primary deficit.
In research terms, this mechanistic distinction allows investigators to decompose the sexual arousal response into its central (motivational, desire, central autonomic activation) and peripheral (vascular, genital engorgement) components. Using PT-141 alongside a PDE5 inhibitor in a factorial experimental design allows separate quantification of each component’s contribution to overall arousal function — an experimental approach that is not possible with either compound class alone.
| ★ | Research note: PT-141 produced statistically significant improvements in desire scores in the HSDD trials, even in women without identifiable peripheral vascular impairment — populations where PDE5 inhibitors have not shown equivalent efficacy in HSDD research. This finding is consistent with PT-141’s mechanism, targeting the central desire/motivation component rather than peripheral arousal capacity, and it illustrates how mechanism-specific research compounds can identify distinct anatomical contributions to sexual function. |
Kisspeptin Research and the HPG Axis: Mechanisms of Reproductive Regulation

Kisspeptin Pulse Characteristics and LH Response
One of the most well-replicated findings in kisspeptin research is the consistency of the LH pulse triggered by exogenous kisspeptin administration across multiple species and across both sexes. In the Dhillo et al. (2005) trial — the first controlled human kisspeptin administration study — healthy male volunteers receiving intravenous kisspeptin-54 showed dose-dependent increases in plasma LH within 15–30 minutes, peaking at approximately 45–60 minutes and returning toward baseline by 90 minutes. This time course corresponds to a single GnRH pulse followed by pituitary LH release and subsequent GnRH receptor resensitization — exactly the dynamics expected from the known physiology of pulsatile GnRH secretion.
The LH response to kisspeptin is sensitive to the gonadal steroid milieu, which provides information about the feedback status of the HPG axis at the time of measurement. In hypogonadal men with low testosterone (where negative feedback is reduced), the LH response to kisspeptin is exaggerated relative to eugonadal men — reflecting the increased sensitivity of GnRH neurons to kisspeptin drive when sex steroid suppression of GnRH neuron activity is removed. This feedback-dependent kisspeptin sensitivity has been used in research as a probe for HPG axis suppression status — comparing pre- and post-treatment kisspeptin LH responses to quantify the degree of HPG axis activation achieved by an intervention.
Kisspeptin in Female Reproductive Biology
Female reproductive biology involves kisspeptin at two distinct levels. At the ARC, kisspeptin/KNDy neurons generate the tonic GnRH pulsatility that drives baseline LH/FSH secretion and follicular development. At the AVPV, kisspeptin neurons mediate the preovulatory LH surge in response to the positive feedback of estradiol at the end of the follicular phase. These two populations are regulated differently — ARC neurons are suppressed by sex steroids (providing negative feedback), while AVPV neurons are stimulated by estradiol (providing positive feedback in the preovulatory context) — a duality that requires careful consideration when interpreting kisspeptin research in cycling female models.
The IVF ovulation trigger data from Jayasena et al. (2014) demonstrated that kisspeptin could substitute for hCG in triggering oocyte maturation by activating the endogenous GnRH/LH surge mechanism rather than pharmacologically substituting for LH. This approach produced the expected LH surge with reduced duration and amplitude compared to exogenous hCG, which is hypothesized to reduce the risk of OHSS — a potentially dangerous complication of IVF in which excessive LH-like stimulation causes ovarian hyperstimulation. Whether this advantage is confirmed in larger trials remains an active research question.
For research on hormone optimization across sex-related peptides and the HPG axis, see our Hormone Optimization Peptides research overview.
Kisspeptin, Puberty, and Developmental Neuroendocrinology
Kisspeptin’s role in puberty initiation has been established through both human genetics and animal models. In rodents, kisspeptin mRNA expression in the hypothalamus increases dramatically at the onset of puberty, and kisspeptin administration can advance pubertal onset in prepubertal animals. Conversely, kisspeptin antagonism delays puberty. In humans, the activation of kisspeptin neurons is now understood to be one of the primary triggers of the pubertal GnRH activation that drives sexual maturation — a finding that emerged directly from the hypogonadotropic hypogonadism genetics (KISS1R loss-of-function = failure of puberty).
The metabolic regulation of kisspeptin is also a research area with broad implications. ARC kisspeptin neurons express receptors for leptin, insulin, and other metabolic hormones, and are suppressed by energy deficit (low leptin). This is the proposed mechanism by which low body weight — as in anorexia nervosa or high-volume athletic training — suppresses the HPG axis and causes functional hypothalamic hypogonadism. Kisspeptin administration in energy-deficient animal models restores LH pulsatility even when the energy deficit itself is maintained, demonstrating that the GnRH neurons retain functional competence and that it is specifically the loss of kisspeptin drive (from suppressed ARC neurons) that causes the axis suppression.
Research Evidence: Synthesis and Current State
PT-141: The Most Clinically Validated Sexual Health Research Peptide
PT-141/bremelanotide has the most extensive and rigorous evidence base of any compound in the sexual health peptide research category. The FDA approval pathway required demonstration of efficacy in randomized, placebo-controlled Phase 3 trials with pre-specified primary endpoints in a defined patient population — a standard that very few research peptides have met. The two pivotal Phase 3 trials (RECONNECT 1 and RECONNECT 2) enrolled a combined approximately 1,200 premenopausal women with HSDD and demonstrated statistically significant improvements in both satisfying sexual events and sexual desire, with an acceptable safety profile documented over 12 months of intermittent use.
The research implications of the RECONNECT trials go beyond simple efficacy confirmation. The trial design — using standardized sexual event diaries and validated desire questionnaires as endpoints — established a methodological framework for quantifying sexual arousal and desire that is applicable in non-pharmaceutical research contexts. The dose-response characterization, pharmacokinetic data, and adverse event profile from the pharmaceutical development program provide a well-documented context for PT-141 research use that does not exist for most research peptides.
Kisspeptin: Genetically Validated Mechanism, Robust Human Data
Kisspeptin’s evidence base is distinctive for two reasons: it has a genetically validated mechanism (KISS1R loss-of-function causes hypogonadotropic hypogonadism in humans, establishing non-redundant necessity) and it has multiple controlled human administration studies published in high-impact clinical endocrinology journals. The combination of genetic validation and clinical pharmacology data positions kisspeptin among the best-characterized research peptides in the neuroendocrine category — comparable in evidence quality to DSIP’s deficit and superior to most other peptides in the broader catalog.
The Dhillo group’s sequential work — characterizing LH response pharmacokinetics in healthy males, demonstrating HPG axis rescue in functional hypothalamic amenorrhea, characterizing sex steroid feedback effects on kisspeptin sensitivity, and testing kisspeptin as an IVF trigger — represents a methodologically coherent research program that has progressively validated the compound’s mechanism across increasingly applied experimental contexts. Independent replication of the core kisspeptin → LH pulse findings has been published by multiple groups in multiple countries, which is the standard marker of a research finding’s reliability.
Areas Where Evidence Remains Limited
Despite the strong evidence for both compounds at their respective targets, several research questions remain open. For PT-141, the long-term HPG axis effects of chronic melanocortin receptor stimulation are not fully characterized; the male data, while mechanistically sound, are less extensive than the female data from the HSDD trials; and the relationship between subjective desire measures and objective physiological arousal endpoints requires further research to characterize the degree of their independence. For Kisspeptin, the optimal administration protocol for HPG axis rescue in different hypogonadal conditions is not established; the species-specific differences in AVPV kisspeptin function between rodents and primates introduce translational caution in applying rodent reproductive data to human HPG biology; and the long-term safety of sustained kisspeptin administration in the context of reproductive axis modulation is not documented.
Research-Grade Quality for Sexual Health Peptides
Sexual health peptide research involves CNS-active compounds (PT-141) and reproductive axis-modulating peptides (Kisspeptin), where analytical purity directly determines the reliability of pharmacological and endocrinological endpoints. For LH pulsatility research using Kisspeptin, contamination with a peptide fragment that retains partial KISS1R affinity but lower efficacy would produce an attenuated LH response indistinguishable from reduced GnRH neuron sensitivity — a source of experimental error that compromises mechanistic interpretation.
The quality requirements for sexual health research peptides are:
- HPLC purity ≥98%: Kisspeptin is available in multiple length variants (Kp-10, Kp-54), and the C-terminal RF-amide is required for KISS1R activation. HPLC confirmation that the intended variant accounts for ≥98% of UV-absorbing material is the minimum for ensuring receptor binding specificity. PT-141 is a cyclic heptapeptide, and HPLC confirms that the cyclized form predominates over linear synthesis intermediates.
- Mass spectrometry confirmation: For Kisspeptin-54 (MW ~6,267 Da), ESI-MS or MALDI-TOF confirms full-length synthesis. For the shorter variants (Kp-10, MW ~1,302 Da), mass confirmation is straightforward by ESI. For PT-141 (MW ~1,025 Da as free acid), mass confirmation should document the cyclic peptide molecular weight, which differs from the linear precursor by one water molecule (18 Da).
- CoA documentation: Batch-specific certificate required for LH pulsatility research where inter-experiment reproducibility depends on consistent receptor binding activity.
- Endotoxin testing: For in vivo studies measuring LH/FSH/testosterone as endpoints, endotoxin contamination is a direct confound — LPS activates the immune-neuroendocrine interface to suppress GnRH pulsatility through cytokine-mediated mechanisms. LAL endotoxin testing below the defined limits is warranted for any Kisspeptin in vivo protocol.
Frequently Asked Questions
Q: What is the mechanistic difference between PT-141 and PDE5 inhibitors like sildenafil?
PT-141 acts centrally at hypothalamic MC4R and limbic MC3R receptors, activating an oxytocin-mediated pathway that initiates the spinal cord autonomic signal driving genital vascular response. Sildenafil and other PDE5 inhibitors act peripherally in vascular smooth muscle by blocking cGMP degradation, amplifying the NO-mediated vascular relaxation that has already been initiated by sexual stimulation. PT-141 can initiate the central arousal signal without requiring prior peripheral stimulation; PDE5 inhibitors amplify peripheral vascular response but cannot initiate the arousal drive. In research models where central desire or motivation is the deficit — rather than peripheral vascular capacity — PT-141 is pharmacologically active where PDE5 inhibitors are not. The HSDD clinical trials, conducted in women without measurable peripheral vascular pathology, demonstrated this mechanistic distinction: PT-141 produced significant desire score improvements in a population where PDE5 inhibitors have not shown consistent efficacy.
Q: How does Kisspeptin regulate GnRH pulse generation at the molecular level?
Kisspeptin binds KISS1R — a Gq-coupled GPCR on GnRH neuron dendrites — producing IP3-mediated calcium release from intracellular stores that depolarizes the GnRH neuron and triggers a GnRH pulse. The upstream oscillator that generates the approximately 60–90 minute pulse rhythm in adult humans is the KNDy neuron network in the arcuate nucleus: NKB (released by one KNDy neuron) acts on neighboring KNDy neurons’ NK3 receptors to propagate the kisspeptin pulse initiation signal, while dynorphin A (also released by KNDy neurons) acts at kappa-opioid receptors on the same cells to terminate the kisspeptin pulse after a defined duration. This auto-regulatory NKB → synchronize → dynorphin → silence → NKB oscillator is the proposed molecular clock of GnRH pulse generation, with kisspeptin as its effector output onto GnRH neurons.
Q: Does PT-141’s mechanism differ between males and females?
The receptor pharmacology is identical across sexes — MC4R and MC3R are expressed in the same hypothalamic and limbic regions in both males and females, and the downstream oxytocin-mediated spinal cord activation mechanism operates in both sexes. The peripheral anatomy of the effector response differs (corpus cavernosum in males, clitoral and vaginal vasculature in females), but both are ultimately activated by the same central autonomic cascade. What differs in the published research is the depth of clinical documentation: the pharmaceutical development program prioritized female HSDD as the indication, producing Phase 3 randomized controlled trial data in premenopausal women that does not have an equivalent in males. Male data from preclinical models and small human trials are mechanistically consistent with the female data, but the evidence base is less developed in terms of controlled study design.
Q: What is functional hypothalamic amenorrhea, and why is Kisspeptin research relevant to it?
Functional hypothalamic amenorrhea (FHA) is HPG axis suppression resulting from energy deficit, exercise-induced metabolic stress, or psychological stress — a functional (reversible) form of hypogonadotropic hypogonadism without structural pathology. The mechanism is suppression of ARC kisspeptin neurons by low leptin (energy deficit signal) or elevated cortisol (stress signal), which reduces kisspeptin drive to GnRH neurons and thereby reduces GnRH pulsatility, LH/FSH, and gonadal steroid production. Kisspeptin research in FHA is relevant because it allows researchers to test whether GnRH neuron responsiveness is preserved even when endogenous kisspeptin input is suppressed, as Jayasena et al. (2009) demonstrated it is. This finding has mechanistic and translational implications: it establishes FHA as a kisspeptin-input deficiency rather than a GnRH neuron competence failure, which changes the research questions that can be asked about HPG axis restoration in this condition.
Q: What quality specifications should a research protocol document for Kisspeptin LH pulsatility studies?
For LH pulsatility studies using Kisspeptin, the protocol should specify: the Kisspeptin variant being used (Kp-10 or Kp-54, as they have different pharmacokinetics and potency profiles in published models); HPLC purity ≥98% with UV trace documentation; ESI-MS or MALDI-TOF molecular weight confirmation; endotoxin level below a defined limit (recommend <0.1 EU/mg for in vivo studies measuring hormonal endpoints); batch-specific CoA with synthesis date; and reconstitution conditions including pH and vehicle. Given that LH pulse amplitude is a primary outcome in many Kisspeptin studies, documenting the basal LH pulsatility of research subjects before intervention — using high-frequency blood sampling with validated LH pulse detection algorithms (Deconvolution, iHormone) — is a methodological requirement for interpretable results.
Conclusion
Sexual health peptide research covers two distinct levels of the reproductive neuroendocrine hierarchy. PT-141’s melanocortin receptor mechanism addresses the central arousal and desire circuitry directly — in the hypothalamic PVN, the limbic system’s MPOA and nucleus accumbens — and has the most extensively documented human clinical evidence base of any compound in the sexual health peptide category, including FDA approval data from Phase 3 randomized controlled trials. Kisspeptin operates at the master-regulator level of the HPG axis, where KISS1R activation on GnRH neurons drives the pulsatile gonadotropin release that determines gonadal steroid production and gametogenesis. Its genetically validated mechanism and multiple controlled human administration studies make it the best-characterized research tool for studying HPG axis biology in pharmacological research settings.
Spartan Peptides supplies PT-141 and Kisspeptin through the Sexual Health research catalog at ≥98% HPLC-verified purity with full CoA documentation. For research spanning the hormone optimization context, see our Hormone Optimization Peptides research overview.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
Dhillo, W.S. et al. (2005). Kisspeptin-54 stimulates the hypothalamic-pituitary gonadal axis in male rats. Journal of Clinical Endocrinology & Metabolism, 90(12), 6609–6615.
Jayasena, C.N. et al. (2009). Kisspeptin-54 triggers egg maturation in women undergoing in vitro fertilization. Journal of Clinical Investigation, 124(8), 3667–3677.
Jayasena, C.N. et al. (2009). Subcutaneous infusion of kisspeptin-54 stimulates gonadotropin release in women, and the response is modulated by sex steroid feedback. Journal of Clinical Endocrinology & Metabolism, 96(11), E1963–E1972.
Clayton, A.H. et al. (2016). Bremelanotide for female sexual dysfunctions in premenopausal women: a randomized, placebo-controlled dose-finding trial. Women’s Health, 12(3), 325–337.
Simon, J.A. et al. (2019). Efficacy and safety of intranasal bremelanotide for hypoactive sexual desire disorder among premenopausal women: a randomized, placebo-controlled trial. JAMA Internal Medicine, 179(10), 1471–1480.
Rosen, R.C. et al. (1997). The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. Journal of Sex & Marital Therapy, 26(2), 191–208.
Seminara, S.B. et al. (2003). The GPR54 gene is a regulator of puberty. New England Journal of Medicine, 349(17), 1614–1627.
Navarro, V.M. (2020). New Insights into the Control of Pulsatile GnRH Release: The Role of Kiss1/Neurokinin B Neurons. Frontiers in Endocrinology, 11, 592109.
Spartan Research Library
Go Deeper on the Science
Browse study indexes, compound comparisons, and protocol stacks in the Spartan Peptides Research Library.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →