Peptide Fundamentals: What Are Peptides, How They Work, and Why They Matter
Written bySpartan Research Team
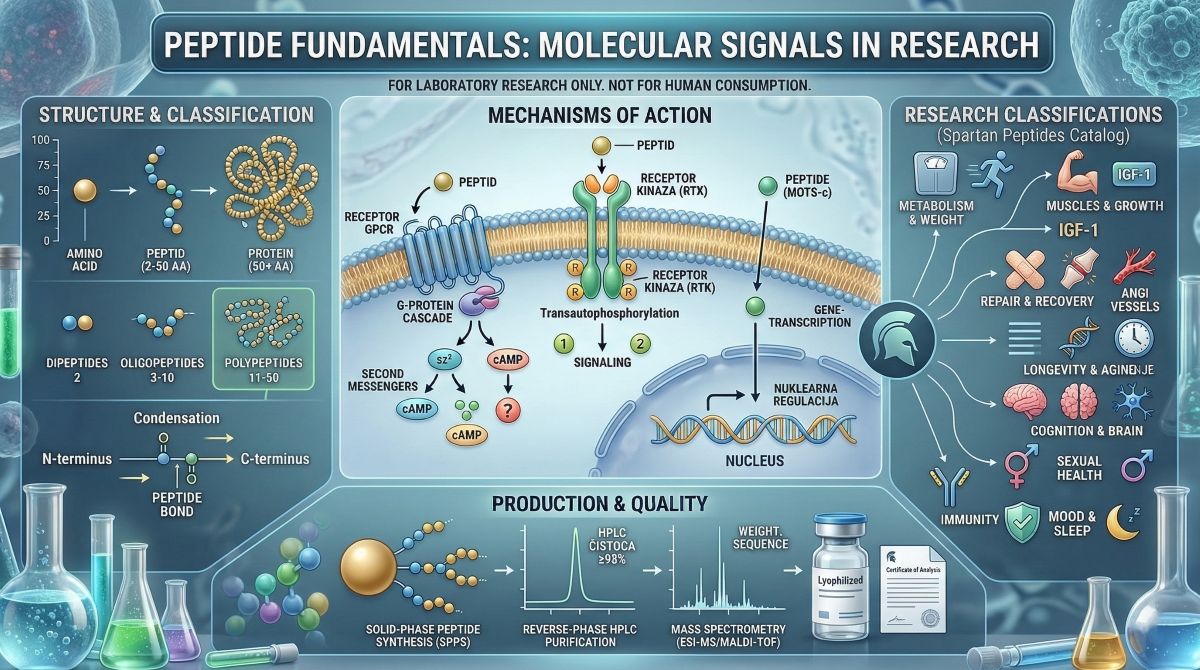
Peptides are short chains of amino acids that function as molecular signals in virtually every biological system studied in modern research. The question of what peptides are has a precise biochemical answer, but the research interest in these molecules extends well beyond their structural definition. Over the past three decades, peptides have become central tools in pharmacology, endocrinology, metabolic research, and tissue biology — not because they are new molecules, but because advances in solid-phase peptide synthesis and analytical chemistry have made it possible to produce and characterize them at research-grade purity for use in controlled laboratory settings.
This guide covers peptide basics from the ground up: what peptides are at the chemical level, how they differ from proteins and small molecules, how they act on biological systems through receptor binding and intracellular signaling, and how the field of peptide research is currently organized by biological function. It also addresses the practical requirements of peptide research — synthesis, purity standards, storage, and reconstitution — and maps Spartan Peptides’ catalog to the research areas where each compound class has documented scientific activity.
All compounds discussed here are research peptides supplied strictly for laboratory investigation. They are not approved for human consumption and are referenced exclusively in the context of scientific research.
What Are Peptides? Biochemistry and Structural Definition
A peptide is a molecule composed of two or more amino acids linked by peptide bonds. A peptide bond forms through a condensation reaction between the carboxyl group (-COOH) of one amino acid and the amino group (-NH2) of the next, releasing a water molecule and producing an amide linkage (-CO-NH-). The resulting chain has a free amino terminus (N-terminus) at one end and a free carboxyl terminus (C-terminus) at the other, which defines its directionality — a convention always written N to C.
Peptides are classified by chain length:
- Dipeptides: 2 amino acids
- Oligopeptides: 3–10 amino acids
- Polypeptides: 11–50 amino acids (the range where most research peptides fall)
- Proteins: 50+ amino acids, typically with a defined three-dimensional structure
The boundary between polypeptides and proteins is not rigid — some sources place it at 100 amino acids, others at defined structural criteria — but for practical research purposes, the distinction matters primarily because peptides of under ~50 residues can be chemically synthesized with high precision and purity, while proteins generally require recombinant expression systems. This is what makes peptides particularly useful as research tools: they can be manufactured to exact sequence specifications, modified to improve stability or bioavailability, and characterized by straightforward analytical methods.
Amino Acid Building Blocks
Twenty standard proteinogenic amino acids serve as the building blocks for all naturally occurring peptides and proteins, each defined by its side chain (R group). Side chain properties govern the peptide’s chemical behavior: charged residues (Asp, Glu, Lys, Arg, His) influence solubility and receptor binding at physiological pH; hydrophobic residues (Phe, Val, Ile, Leu, Trp) drive membrane interactions and hydrophobic core packing; polar uncharged residues (Ser, Thr, Gln, Asn, Tyr) participate in hydrogen bonding with targets.
Research peptides are sometimes synthesized with non-proteinogenic amino acids or D-amino acid substitutions. D-amino acids — the mirror-image configuration of the natural L-form — resist proteolytic degradation, since most mammalian peptidases are stereospecific for L-amino acids. This modification extends plasma half-life and is used in several research compounds to improve their stability for in vivo applications.
How Peptides Differ from Small Molecules and Proteins
Peptides occupy a pharmacological space between small molecules (MW < 500 Da, typically) and full-sized proteins (MW > 10,000 Da). This positioning has practical research consequences. Small molecules generally cross cell membranes freely and have long shelf stability, but their mechanism of action is often broad — hitting multiple molecular targets, which complicates interpretation in research settings. Proteins have high target specificity but poor membrane permeability, immunogenicity risk, and manufacturing complexity.
Peptides of 5–50 residues can combine target specificity approaching that of proteins with manufacturing precision approaching that of small molecules. Their typical MW range (500–6,000 Da) places them at the upper end of what can be fully characterized by mass spectrometry, and their sequences can be confirmed unambiguously by tandem MS fragmentation — a quality control advantage over larger biologics.
| ★ | Research note: The term ‘research peptide’ refers specifically to synthetic peptides produced for laboratory investigation, characterized by HPLC purity ≥98% and mass spectrometry sequence confirmation, and supplied with a batch-specific Certificate of Analysis. This distinguishes them from pharmaceutical-grade peptides (which require GMP manufacturing) and dietary amino acid supplements (which lack analytical purity documentation). |
How Peptides Work: Receptor Biology and Signaling Mechanisms

Peptides exert their biological effects primarily through receptor binding — interacting with specific protein receptors on cell surfaces or, less commonly, intracellularly. The specificity of peptide-receptor interactions is the defining characteristic that distinguishes peptide pharmacology from small-molecule pharmacology: a peptide typically binds one receptor class (or a small number of closely related receptors), whereas many small molecules interact with multiple targets.
G Protein-Coupled Receptors (GPCRs)
The largest class of peptide receptors is the G protein-coupled receptor (GPCR) superfamily — a group of seven-transmembrane-domain receptors that transduce extracellular signals into intracellular second messenger cascades. Approximately 800 GPCRs are encoded in the human genome, and a substantial fraction are receptors for endogenous peptides: the GLP-1 receptor, ghrelin receptor (GHSR), GnRH receptor, oxytocin receptor, and many others are all GPCRs whose natural ligands are peptides.
When a peptide binds its GPCR, conformational changes in the receptor allow coupling to heterotrimeric G proteins (Gs, Gi, Gq, and others), which in turn activate or inhibit downstream effectors: adenylyl cyclase (cAMP production), phospholipase C (DAG/IP3 production), or ion channels. The specific G protein coupling determines whether the signal is excitatory or inhibitory and which second messengers are produced. This receptor-level specificity is why peptides targeting the same GPCR can have fundamentally similar biological effects regardless of their structural differences.
Receptor Tyrosine Kinases (RTKs)
Several peptide growth factors act through receptor tyrosine kinases — single-pass transmembrane receptors whose intracellular domain has intrinsic kinase activity. Insulin, IGF-1, EGF, and PDGF all signal through RTKs. When a peptide ligand binds, it induces receptor dimerization and transautophosphorylation of tyrosine residues in the intracellular domain. These phosphotyrosines serve as docking sites for adapter proteins (Grb2, IRS-1, Src, etc.) that recruit downstream kinases, including PI3K and MAPK cascades. RTK-mediated signaling governs cell proliferation, survival, differentiation, and metabolism, making it a target for both growth factor research and oncology.
Nuclear Receptors and Intracellular Mechanisms
Some peptide-derived compounds act through mechanisms that bypass cell-surface receptors entirely. MOTS-c, a mitochondria-derived peptide, translocates to the nucleus under metabolic stress, where it directly regulates gene transcription. This intracellular mechanism is distinct from classical receptor pharmacology and represents an emerging class of peptide function that operates at the level of gene regulation rather than second messenger signaling.
Copper peptides such as GHK-Cu interact with intracellular signaling networks through copper coordination chemistry, activating gene expression programs related to collagen synthesis, antioxidant defense, and tissue remodeling. The copper ion is not incidental — it is required for the biological activity of the complex, participating in enzymatic reactions including lysyl oxidase-mediated cross-linking of newly synthesized extracellular matrix.
Half-Life, Degradation, and Bioavailability
Endogenous peptides are typically short-lived in plasma. GLP-1 has a native half-life of under two minutes, degraded by dipeptidyl peptidase-4 (DPP-4). Substance P and other neuropeptides are cleared within minutes. This rapid degradation is physiologically appropriate for signaling molecules that need to act acutely and locally; it becomes a challenge for research peptides that need to maintain receptor activity over the duration of an experiment.
Research peptides address this through several engineering approaches: albumin-binding moieties (as in CJC-1295), DPP-4 cleavage site protection, D-amino acid substitution, PEGylation, and fatty acid conjugation (as in semaglutide). The practical research consequence is that different compounds in the same mechanistic class can have half-lives ranging from hours to days — a variable that must be controlled in study design. Oral bioavailability is a separate challenge: most peptides above ~10 residues are degraded in the gastrointestinal tract before absorption, which is why research protocols typically use parenteral administration routes.
Peptide Research Classification by Biological Function
Research peptides are most usefully organized by the biological systems they target. The following classification maps directly to the research areas covered across Spartan Peptides’ catalog:
Metabolic and Weight Regulation Peptides
GLP-1 receptor agonists (semaglutide analogues), dual GIP/GLP-1 agonists (tirzepatide analogues), triple agonists (retatrutide), growth hormone-releasing hormones, and direct lipolytic fragments (AOD-9604) are all studied for their effects on energy balance, adipose tissue biology, and metabolic regulation. The weight loss research catalog covers this compound class. The mechanism, receptor targets, and key study data for each are reviewed in the Complete Guide to Weight Loss Peptides.
Muscle Development and Growth Hormone Axis Peptides
GHRH analogues (CJC-1295, tesamorelin) and GHRPs (ipamorelin) stimulate pulsatile growth hormone secretion from the anterior pituitary through GHRH receptors and ghrelin receptors, respectively. Their research relevance spans body composition, lean mass maintenance, and IGF-1-mediated anabolic signaling in skeletal muscle.
Repair and Recovery Peptides
BPC-157, TB-500 (Thymosin Beta-4), and related compounds target angiogenesis, extracellular matrix remodeling, fibroblast activation, and cell migration in injured tissue. Their preclinical evidence base covers tendon, ligament, muscle, gut mucosa, and peripheral nerve repair models.
Anti-Aging and Longevity Peptides
NAD+ (for sirtuin/PARP-mediated metabolic and DNA repair pathways), Epithalon (for telomerase activation and pineal melatonin regulation), MOTS-c (for AMPK-mediated mitochondrial signaling), and GHK-Cu (for ECM remodeling and antioxidant gene expression) represent four distinct mechanisms studied in the context of cellular aging.
Cognitive and Nootropic Peptides
Semax, Selank, Dihexa, and Pinealon are peptides studied for their effects on neurogenesis, BDNF signaling, cognitive performance, and neuroprotection.
Semax is a synthetic analogue of ACTH(4-10) that has been documented to increase BDNF and NGF expression in rodent CNS models; Selank is a heptapeptide with anxiolytic and nootropic effects attributed to GABAergic modulation.
The focus and clarity research catalog includes Semax and Pinealon.
Sexual Health Peptides
PT-141 (bremelanotide) is a melanocortin receptor agonist — specifically MC3R and MC4R — whose activation in the hypothalamus modulates sexual arousal through central nervous system pathways rather than vascular mechanisms.
Kisspeptin is a neuropeptide that acts upstream of GnRH to regulate reproductive hormone release, with documented effects on LH pulsatility and gonadal function in research models.
Immunity Peptides
Thymosin Alpha-1 is a 28-amino acid peptide derived from the thymus gland that modulates innate and adaptive immune responses through Toll-like receptor signaling, dendritic cell maturation, and T-cell activation pathways. It has been the most clinically studied thymosin family peptide, with trials in chronic hepatitis B and C, cancer, and sepsis.
Mood and Sleep Peptides
DSIP (Delta Sleep-Inducing Peptide) is a nonapeptide that modulates sleep architecture through effects on slow-wave sleep. Selank’s anxiolytic profile, which involves modulation of enkephalin metabolism and GABA-A receptor activity, positions it in this category as well.
How Research Peptides Are Made: Synthesis and Characterization
Solid-Phase Peptide Synthesis (SPPS)
The majority of research peptides are produced by solid-phase peptide synthesis (SPPS), a method developed by Robert Bruce Merrifield in the 1960s for which he received the Nobel Prize in Chemistry in 1984. In SPPS, the C-terminal amino acid is attached to a solid resin support, and the peptide chain is assembled stepwise from C-terminus to N-terminus by sequential addition of protected amino acid building blocks (Fmoc or Boc chemistry). After each coupling, deprotection removes the temporary protecting group on the incoming residue, freeing it for the next coupling.
The advantages of SPPS are precision and scalability: every residue is added in a defined order with monitored coupling efficiency, and the resin support simplifies purification by allowing wash steps between each cycle. Modern automated SPPS synthesizers can assemble peptides of 30–50 residues in a matter of days with high fidelity, though longer sequences accumulate deletion and truncation errors that require stringent purification to remove.
After assembly and cleavage from the resin, the crude peptide mixture is purified by reverse-phase high-performance liquid chromatography (RP-HPLC) to remove synthesis byproducts. The purified compound is then lyophilized (freeze-dried) to produce the powder form in which research peptides are typically supplied.
Key Quality Metrics: HPLC, Mass Spectrometry, CoA
Three analytical methods define the quality of a research peptide:
- HPLC purity analysis: Reverse-phase HPLC separates the target compound from synthesis impurities based on hydrophobicity. UV detection at 214–220nm (where the peptide bond absorbs) quantifies the fraction of total UV-absorbing material attributable to the target peak. A purity of ≥98% means the main peak accounts for at least 98% of this signal. This is the standard minimum for research-grade peptides and the most widely cited purity metric in the research literature.
- Mass spectrometry confirmation: Electrospray ionization (ESI-MS) or matrix-assisted laser desorption/ionization (MALDI-TOF) determines the molecular weight of the compound to within fractions of a Dalton. For a peptide with a known sequence, the theoretical molecular weight can be calculated precisely; a measured mass matching the theoretical value confirms correct sequence composition. Tandem MS (MS/MS) fragmentation can additionally confirm the sequence of individual residues, which is particularly important for longer peptides where positional errors might not be detected by mass alone.
- Certificate of Analysis (CoA): A batch-specific document reporting the actual HPLC purity percentage, the mass spectrometry data, synthesis date, and compound identity. The CoA is the primary traceability document in peptide research — it links a specific vial of compound to the analytical data that characterize its quality. Any research protocol requiring reproducibility should document the CoA data for every compound used.
Storage, Stability, and Reconstitution
Lyophilized peptide powders are generally stable at -20°C for one to two years when stored dry, away from light, and protected from humidity. The primary degradation pathways for stored peptides are hydrolysis (water-mediated peptide bond cleavage, accelerated by residual moisture), oxidation (particularly of methionine, cysteine, and tryptophan residues), and aggregation (intermolecular association that removes the compound from solution).
Reconstitution — dissolving a lyophilized peptide powder in an aqueous vehicle for use in research — requires attention to solubility characteristics that vary by sequence. Acidic peptides (net negative charge) often dissolve best in mildly basic aqueous solutions; basic peptides dissolve best in dilute acid; hydrophobic peptides may require initial dissolution in a small volume of acetonitrile or DMSO before aqueous dilution. Detailed reconstitution protocols by peptide class are covered in our Peptide Reconstitution and Storage Guide.
Peptide Research: Study Design and Evidence Standards
In Vitro, Ex Vivo, and In Vivo Research
Peptide research progresses through a hierarchy of experimental systems with increasing biological complexity and translational relevance. In vitro studies use cell cultures — receptor-expressing cell lines for binding assays, primary cells for mechanistic studies — to characterize receptor binding, signaling pathway activation, and direct cellular effects. These studies offer precise experimental control and are well-suited for mechanism characterization, but cells in culture lack the physiological context (blood supply, hormonal milieu, tissue architecture) that governs peptide behavior in a living system.
Ex vivo studies use tissues or organs removed from animals — perfused hearts, isolated tendons, tissue slices — that retain more physiological complexity than cell culture while still allowing experimental manipulation not possible in a live animal. In vivo studies in rodents, primates, or other animal models provide the full pharmacokinetic and pharmacodynamic context: absorption, distribution, metabolism, excretion, and systemic effects are all operating simultaneously. The limitation is species translation — rodent physiology differs from human physiology in ways that are sometimes predictable and sometimes not.
Human clinical trials sit at the top of the evidence hierarchy, but most research peptides have not undergone the formal pharmaceutical development process that would produce Phase 1, 2, or 3 human data. The majority of the published peptide research literature is therefore preclinical, which is a standard feature of early-stage research compounds rather than an indication of safety concern.
Peptide Stacking and Combination Research
Research protocols increasingly examine combinations of peptides from mechanistically distinct classes, reflecting the observation that many biological processes are regulated by multiple pathways simultaneously. A GH secretagogue combined with a connective tissue repair peptide addresses both anabolic signaling and the structural substrate for mechanical loading; a mitochondrial-targeted peptide combined with a NAD+ precursor addresses mitochondrial function from two independent upstream entry points.
The Legal and Regulatory Status of Research Peptides
Research peptides occupy a specific regulatory category: they are not classified as FDA-approved drugs, not regulated as dietary supplements, and not scheduled as controlled substances (with a small number of exceptions). In the United States, research compounds can be legally manufactured and supplied for laboratory research use, provided they are not labeled or sold for human consumption. This regulatory framework governs how research peptide suppliers operate and how researchers can legitimately procure and use these compounds.
Where the Peptide Research Evidence Is Strongest

Not all peptide research areas have equivalent evidence bases. Understanding which compound classes have the most developed science — and where the data remains early-stage — is necessary for calibrating research expectations.
Most Developed Evidence: GLP-1 Agonist Class
The GLP-1 receptor agonist class has the most extensive human clinical trial data of any research peptide category, driven by the pharmaceutical development of semaglutide and tirzepatide for type 2 diabetes and obesity indications. Phase 3 randomized controlled trials with thousands of participants, 68–72 week follow-up periods, and pre-specified primary endpoints provide the highest-quality evidence in the peptide research literature. The clinical data are supplemented by extensive mechanistic work characterizing GLP-1 receptor pharmacology, central nervous system effects, and cardiovascular outcomes.
Strong Preclinical Evidence, Limited Human Data: BPC-157, TB-500
BPC-157 has more published preclinical studies than almost any other research peptide — consistent findings across more than 50 published papers spanning multiple tissue types, multiple injury models, and multiple research groups. The breadth and consistency of these preclinical data are what sustain research interest despite the absence of controlled human clinical trials. TB-500 (Thymosin Beta-4) has a more advanced human context through clinical trials in ophthalmic indications and cardiac applications, though not in the musculoskeletal applications most studied preclinically.
Emerging Data: MOTS-c, Epithalon, Cognitive Peptides
MOTS-c, Epithalon, and the cognitive research peptide class (Semax, Selank, Dihexa) have smaller but methodologically sound evidence bases. MOTS-c research from high-impact journals (Cell Metabolism, 2015; Nature Aging, 2022) establishes strong mechanistic credibility. Epithalon’s evidence spans several decades but comes predominantly from a single research group. Cognitive peptides have a longer Russian research history, with more recent independent investigation confirming core findings. In each case, the appropriate scientific posture is to recognize what the evidence shows and what it does not yet demonstrate.
| ★ | Research principle: The strength of a compound’s evidence base does not necessarily correlate with its mechanistic plausibility. Some well-characterized peptides lack large clinical trial programs because they have not entered pharmaceutical development, not because the underlying biology is uncertain. Evaluating peptide research requires distinguishing mechanistic evidence from clinical outcome data. |
Sourcing Research Peptides: What to Look For
The quality of research outcomes in peptide biology is directly dependent on the quality of the compounds used. A peptide with sub-98% purity, incorrect molecular weight, or bacterial endotoxin contamination will produce data that cannot be confidently attributed to the target compound — or, in in vivo models, will produce inflammatory confounds that corrupt the biological signal entirely.
When evaluating a research peptide supplier, the minimum documentation required is:
- HPLC purity certificate ≥98% with UV trace showing the main peak and impurity baseline
- Mass spectrometry data (ESI-MS or MALDI-TOF) confirming molecular weight within ±0.1% of theoretical
- Batch-specific CoA with synthesis date and compound identity
- US-origin manufacturing for intact cold-chain management and shorter shipping transit times
- Endotoxin testing data for any compound used in in vivo research models
Frequently Asked Questions
Q: What distinguishes a research peptide from a pharmaceutical peptide?
Research peptides and pharmaceutical peptides share the same fundamental chemistry — both are amino acid chains produced by SPPS — but differ in manufacturing standards, documentation, and intended use. Pharmaceutical peptides (such as insulin, liraglutide, or FDA-approved tesamorelin) are produced under Current Good Manufacturing Practice (cGMP) regulations, which govern facility standards, batch records, stability testing, and sterility. Research peptides are produced to research-grade standards — primarily ≥98% HPLC purity and mass spectrometry confirmation — without the full cGMP manufacturing infrastructure. This makes them appropriate for laboratory research, where the primary requirement is analytical purity and batch traceability, but not for therapeutic use in humans.
Q: Why are peptide research compounds sold as powders rather than in solution?
Lyophilized (freeze-dried) powder is the standard supply form for research peptides because peptide solutions degrade significantly faster than dry powders. In aqueous solution, peptide bonds are subject to hydrolysis, and residues such as methionine, tryptophan, and cysteine are vulnerable to oxidation — processes that proceed rapidly at room temperature and even at refrigerated temperatures over weeks. As dry powder stored at -20°C, most research peptides maintain stability for one to two years. Researchers reconstitute the powder immediately before use, preparing only the volume needed for the current experiment to minimize the time the compound spends in solution.
Q: What is the difference between a peptide and a protein in research contexts?
The distinction is primarily one of size and production method, with practical research consequences. Peptides under approximately 50 amino acids can be synthesized by SPPS with a defined sequence and high analytical purity. Proteins are typically too large for SPPS and are instead produced by recombinant expression in bacterial, yeast, or mammalian cell systems, a process that introduces additional variables, including glycosylation patterns, folding states, and host-cell impurities. For research purposes, peptides have the advantage of being fully analytically characterizable by HPLC and mass spectrometry, while protein characterization requires more complex analytical methods. The boundary is not absolute: some smaller proteins (< 100 residues) can be made by SPPS, while some longer synthetic peptides require native chemical ligation for assembly.
Q: How do researchers verify that a peptide is producing its expected biological effect rather than an artifact?
Mechanistic verification in peptide research typically involves several orthogonal approaches. Receptor binding assays (radioligand displacement, surface plasmon resonance, or FRET-based methods) confirm that the compound binds its expected receptor with the expected affinity. Downstream signaling readouts — second messenger quantification (cAMP, IP3), reporter gene assays, or phosphorylation blots — confirm that binding activates the expected intracellular pathway. Negative controls with receptor antagonists or peptide analogues lacking biological activity are used to confirm specificity. For in vivo studies, dose-response relationships and receptor-knockout animal models provide additional specificity evidence. The combination of binding data, pathway activation data, and functional outcome data in the same model system is the standard for establishing that a biological effect is attributable to the intended mechanism.
Q: What is the significance of peptide purity in research, and why is 98% the standard threshold?
Purity in the research context means that the compound being studied is predominantly (≥98%) the molecule of interest, with impurities accounting for ≤2% of the material. The 98% threshold is a pragmatic standard that balances synthesis economics with research reliability — it is achievable by HPLC purification and sufficient to ensure that observed biological effects are attributable to the target compound rather than trace contaminants. Sub-98% purity becomes problematic when studying dose-response relationships (impurities contribute a biologically active or inactive fraction that skews the apparent potency), when comparing batches (variable impurity profiles produce variable results), or when the compound acts at low concentrations where impurity contributions are proportionally larger. For GH secretagogues acting in the nanomolar range, for example, a 5% impurity might represent a compound that occupies a meaningful fraction of receptors at the research concentration.
Conclusion
Peptides are among the most precise molecular tools available in modern biological research. Their ability to target specific receptors with high selectivity, their amenability to sequence-level modification, and their full characterizability by standard analytical methods make them well-suited for studies ranging from receptor pharmacology to metabolic biology to tissue repair. The peptide research field spans a broad range of biological systems — metabolic regulation, muscle and body composition, cellular aging, tissue repair, cognitive function, sexual health, immune modulation, and sleep biology — each with its own evidence base and mechanistic logic.
Spartan Peptides maintains a research catalog covering all eight of these areas — from GLP-1 agonists and GH secretagogues to repair peptides, longevity compounds, and nootropic peptides — all supplied at ≥98% HPLC-verified purity with batch-specific CoA documentation.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
Merrifield, R.B. (1963). Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide. Journal of the American Chemical Society, 85(14), 2149–2154.
Fosgerau, K. & Hoffmann, T. (2015). Peptide therapeutics: current status and future directions. Drug Discovery Today, 20(1), 122–128.
Uhlen, M. et al. (2015). Proteomics. Tissue-based map of the human proteome. Science, 347(6220), 1260419.
Drucker, D.J. (2018). Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metabolism, 27(4), 740–756.
Raun, K. et al. (1998). Ipamorelin is the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5), 552–561.
Sikiric, P. et al. (2018). Stable Gastric Pentadecapeptide BPC 157 — NO-System Relation. Current Pharmaceutical Design, 24(18), 1990–2001.
Lee, C. et al. (2015). The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metabolism, 21(3), 443–454.
Goldstein, A.L. et al. (2012). Thymosin beta-4: a multi-functional regenerative peptide. Expert Opinion on Biological Therapy, 12(1), 37–51.
Khavinson, V.Kh. et al. (2003). Peptide Epitalon activates telomerase in human somatic cells. Neuro Endocrinology Letters, 24(3–4), 162–165.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →