What Is Semaglutide? GLP-1 Peptide Basics Explained
Written bySpartan Research Team
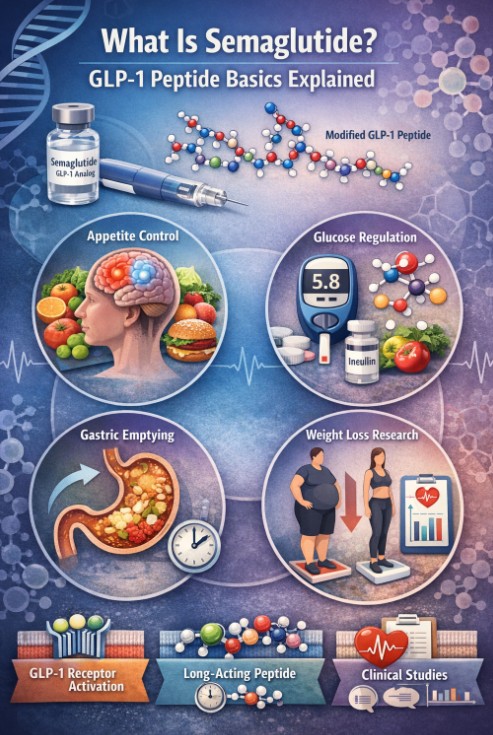
What Is Semaglutide?<\/b><\/h2>
 Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span>
Key Research on Semaglutide<\/b><\/h2>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span>
Key Research on Semaglutide<\/b><\/h2>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span>
Key Research on Semaglutide<\/b><\/h2>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span>
Key Research on Semaglutide<\/b><\/h2>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span>
Key Research on Semaglutide<\/b><\/h2>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>

STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span>
Is semaglutide considered a GLP-1 peptide?<\/b>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span>
How does semaglutide differ from natural GLP-1?<\/b>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span>
Why is semaglutide discussed so often in weight-related research?<\/b>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span>“,”rendered”:”For researchers asking <\/span>what semaglutide is<\/b>, the answer starts with GLP-1 biology. Semaglutide is a long-acting <\/span>GLP-1 research peptide<\/b> studied for its effects on appetite signaling, glucose regulation, gastric emptying, and body-weight outcomes across human and preclinical research models. Published literature describes it as a modified GLP-1 receptor agonist engineered for longer activity than native GLP-1, which is one reason it remains central to metabolic peptide research. In this article, the focus stays on what semaglutide is, how it works, why it is studied, and what the research record shows.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Is Semaglutide a Peptide?<\/b><\/h2>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
Appetite and satiety signaling<\/b><\/h3>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Gastric emptying<\/b><\/h3>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Why prolonged activity matters<\/b><\/h3>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Why Researchers Study Semaglutide<\/b><\/h2>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Pharmacokinetic reviews<\/b><\/h3>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
Research Considerations and Limitations<\/b><\/h2>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
FAQ<\/b><\/h2>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
Conclusion<\/b><\/h2>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
What Is Semaglutide?<\/b><\/h2>
 <\/p>
<\/p>
Semaglutide is a synthetic peptide analog of glucagon-like peptide-1, or GLP-1. In the research literature, it is typically discussed as a GLP-1 receptor agonist, meaning it activates the same receptor targeted by endogenous GLP-1 signaling. Native GLP-1 is rapidly degraded, while semaglutide was designed to remain active much longer in circulation. That longer exposure changed the way researchers could study sustained GLP-1 receptor signaling over time.<\/span><\/p>
A useful internal starting point for Spartan readers is the<\/span> GLP-1 Sema research peptide<\/span><\/a>. For a broader context around related compounds, the article should also connect naturally to the site’s<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
From a peptide-development standpoint, semaglutide is not just another compound in a large catalog. It is a modified GLP-1 analog built to address the short half-life of the native hormone. Reviews of semaglutide pharmacology describe amino-acid substitutions and acylation features that increase albumin binding and slow breakdown, which helps explain the compound’s prolonged activity profile.<\/span><\/p>
GLP-1 stands for glucagon-like peptide-1, an incretin hormone released from intestinal L cells in response to nutrient intake. Research on GLP-1 has focused on several connected pathways: glucose-dependent insulin secretion, glucagon suppression, gastric emptying, and satiety-related signaling. Because these functions span gut, pancreas, and central appetite circuits, GLP-1 biology became one of the main frameworks for modern obesity and metabolic research.<\/span><\/p>
When readers encounter the phrase <\/span>GLP-1 peptide<\/b>, they are often seeing shorthand for one of two things: the native hormone itself, or a modified peptide analog designed to reproduce GLP-1 receptor activation over a longer time frame. Semaglutide belongs to the second group. It follows the biological template of GLP-1 but was engineered for greater persistence, which made it more practical for long-duration research.<\/span><\/p>
Readers who are earlier in the topic often benefit from a broader foundation before going deeper into compound-specific material. That is where an internal supporting link like<\/span> Best Peptides for Weight Loss in 2026<\/span><\/a> fits naturally in the article body.<\/span><\/p>
Yes. <\/span>Semaglutide<\/span><\/a> is a peptide-based research compound. More precisely, it is a modified analog of GLP-1. It is not identical to endogenous human GLP-1, and that distinction matters because the structural differences are what give semaglutide its longer pharmacokinetic profile.<\/span><\/p>
That longer duration comes from deliberate molecular changes rather than from a different receptor target. Research reviews describe semaglutide as maintaining high affinity for the GLP-1 receptor while resisting rapid degradation and showing stronger albumin association in circulation. In peptide science, that makes semaglutide a useful example of how small structural edits can produce large differences in duration of action.<\/span><\/p>
Semaglutide acts through the <\/span>GLP-1 receptor<\/span><\/a>, which is involved in insulin secretion, glucagon regulation, gastric motility, and appetite-related pathways. Once the receptor is activated, downstream signaling affects several systems at once rather than a single isolated endpoint. That is one reason semaglutide studies often report changes in body weight, glucose measures, and gastrointestinal effects within the same trial.<\/span><\/p>
A large part of semaglutide research centers on appetite regulation. GLP-1 signaling is tied to satiety pathways and food-intake regulation, including central nervous system pathways discussed in recent narrative reviews. Researchers have continued to study how GLP-1 receptor agonists interact with central appetite circuits, since that mechanism helps explain why the compound draws such sustained interest in obesity research.<\/span><\/p>
Semaglutide is also studied in relation to delayed gastric emptying. Slower gastric transit can alter postprandial glucose patterns and may contribute to reduced energy intake in study populations. This mechanism appears often in both semaglutide reviews and clinical-trial discussions, especially when authors interpret the compound’s metabolic effects beyond glycemic markers alone.<\/span><\/p>
Native GLP-1 is rapidly inactivated, which limits how long receptor signaling persists. Semaglutide’s design extends that window, allowing researchers to examine cumulative receptor activation over weeks and months rather than over brief physiologic bursts. This longer-acting profile is part of why semaglutide became such a prominent comparator in metabolic peptide research.<\/span><\/p>
For readers <\/span>comparing compounds<\/span><\/a> in the same category.<\/span><\/p>
Semaglutide is studied because it sits at the intersection of appetite biology, body-weight regulation, glucose control, and long-acting peptide design. It matters both as a metabolic research tool and as a template for how native gut hormones can be modified into longer-acting analogs.<\/span><\/p>
One line of research focuses on body-weight outcomes. In the STEP 1 trial, adults with overweight or obesity assigned to weekly semaglutide plus lifestyle intervention showed greater mean body-weight reduction at 68 weeks than placebo-treated subjects. The same trial also reported differences in cardiometabolic risk factors and physical functioning outcomes.<\/span><\/p>
A second line of research concerns obesity with type 2 diabetes. In STEP 2, semaglutide 2.4 mg once weekly produced greater body-weight reduction than placebo in adults with overweight or obesity and type 2 diabetes, again over 68 weeks. This helped show that semaglutide research was not limited to a single population.<\/span><\/p>
A third area is cardiovascular outcomes. In SELECT, investigators studied adults with overweight or obesity and established cardiovascular disease, but without diabetes. The trial reported fewer major adverse cardiovascular events in the semaglutide group than in the placebo group. That result widened the research conversation beyond glucose and body weight alone.<\/span><\/p>
For internal topical coverage, this section can also point readers to<\/span> Top 5 Peptides for Weight Loss in 2025 Update<\/span><\/a> and the<\/span> Weight Loss blog category<\/span><\/a>.<\/span><\/p>
The STEP 1 trial remains one of the landmark semaglutide studies. It enrolled 1,961 adults with overweight or obesity without diabetes and followed them for 68 weeks. The semaglutide group showed a mean body-weight change of -14.9% versus -2.4% with placebo, with higher proportions of participants reaching 5%, 10%, and 15% body-weight reduction thresholds. Gastrointestinal adverse events were more common in the semaglutide group, and discontinuation due to GI events was also higher.<\/span><\/p>
STEP 2 examined adults with overweight or obesity and type 2 diabetes. In that trial, semaglutide 2.4 mg once weekly produced an estimated mean body-weight change of -9.6% versus -3.4% with placebo at week 68. Gastrointestinal adverse events were again more frequent in semaglutide-treated subjects. These findings helped reinforce that the compound’s research profile extended across more than one metabolic subgroup.<\/span><\/p>
SELECT enrolled 17,604 adults with overweight or obesity and established cardiovascular disease, but no history of diabetes. A primary cardiovascular endpoint event occurred in 6.5% of the semaglutide group and 8.0% of the placebo group, corresponding to a hazard ratio of 0.80. Permanent discontinuation due to adverse events was more frequent in the semaglutide arm. In research terms, this was one of the studies that moved semaglutide beyond a narrow weight-management frame and into broader cardiometabolic outcome discussion.<\/span><\/p>
Published pharmacokinetic reviews have also shaped how semaglutide is described in the literature. Those reviews emphasize semaglutide’s long half-life, albumin binding, and design features that distinguish it from native GLP-1. This matters because semaglutide’s research value is not only about endpoint data; it is also about what the molecule represents in peptide engineering.<\/span><\/p>
The most direct difference is duration. Native GLP-1 is quickly degraded and has a short physiologic half-life. Semaglutide was designed to remain active much longer, which changes how researchers can study persistent GLP-1 receptor agonism in longer trials and longitudinal metabolic studies.<\/span><\/p>
There is also a design lesson here. Native hormones often have strong biological activity but limited duration. Peptide analog development tries to preserve receptor activity while extending exposure, improving stability, or changing distribution. Semaglutide is one of the clearest examples of that design strategy, producing a widely studied research compound.<\/span><\/p>
Semaglutide remains important because it has become a reference compound in GLP-1 research. Many newer metabolic peptides are now compared against semaglutide rather than discussed in isolation. That benchmark status comes from both its mechanistic clarity and its large evidence base across body-weight and cardiovascular outcomes.<\/span><\/p>
It also remains central because the research conversation continues to expand. Semaglutide is discussed in relation to adiposity, cardiovascular risk, metabolic endpoints, appetite pathways, and next-generation incretin comparisons. Even when later compounds attract attention, semaglutide often remains the point of reference.<\/span><\/p>
The semaglutide literature is large, but not every result should be generalized without context. Trial populations differ. Some studies focus on overweight or obesity without diabetes, some include type 2 diabetes, and others center on cardiovascular disease. Endpoint selection matters too. A change in body weight is not the same endpoint as a change in cardiovascular-event incidence, and those findings should not be merged into one undifferentiated conclusion.<\/span><\/p>
Tolerability also remains part of the research picture. In both STEP 1 and STEP 2, gastrointestinal adverse events were more frequent with semaglutide than with placebo. That does not invalidate the broader literature, but it does mean any serious discussion of the compound should include both outcomes and study-reported limitations.<\/span><\/p>
What is semaglutide used for in research?<\/b><\/p>
Semaglutide is used in research on GLP-1 receptor activation, appetite signaling, glucose regulation, gastric emptying, body-weight outcomes, and cardiometabolic endpoints. It is also used as a comparator when newer incretin-based compounds are studied, since semaglutide already has a large clinical and pharmacologic literature behind it.<\/span><\/p>
Is semaglutide considered a GLP-1 peptide?<\/b><\/p>
Yes. Semaglutide is a modified peptide analog of GLP-1 and is classified as a GLP-1 receptor agonist. Its structure was designed to preserve receptor activity while extending duration in circulation compared with native GLP-1.<\/span><\/p>
How does semaglutide differ from natural GLP-1?<\/b><\/p>
Natural GLP-1 is an endogenous incretin hormone released after nutrient intake and rapidly degraded in the body. Semaglutide is a modified analog built for much longer activity, which makes it more practical for long-duration metabolic research.<\/span><\/p>
Why is semaglutide discussed so often in weight-related research?<\/b><\/p>
The volume of research is one reason. Large trials such as STEP 1 and STEP 2 reported greater body-weight reduction with semaglutide than with placebo in the studied populations, which made semaglutide a recurring reference point in obesity research.<\/span><\/p>
For readers asking <\/span>what semaglutide is<\/b>, the best answer is that it is a long-acting <\/span>GLP-1 research peptide<\/b> built from the GLP-1 template and studied across appetite, glucose, gastric-motility, body-weight, and cardiovascular research settings. The published record made semaglutide one of the main benchmark compounds in modern metabolic peptide research. Within Spartan’s internal content structure, the next natural step is to review the<\/span> GLP-1 Sema research peptide<\/span><\/a>, browse the broader<\/span> Weight Loss Peptides<\/span><\/a> category.<\/span><\/p>
What Does GLP-1 Mean in Peptide Research?<\/b><\/h2>
Is Semaglutide a Peptide?<\/b><\/h2>
How Semaglutide Works<\/b><\/h2>
GLP-1 receptor activation<\/b><\/h3>
Appetite and satiety signaling<\/b><\/h3>
Gastric emptying<\/b><\/h3>
Why prolonged activity matters<\/b><\/h3>
Why Researchers Study Semaglutide<\/b><\/h2>
Key Research on Semaglutide<\/b><\/h2>
 <\/p>
<\/p>
STEP 1: adults with overweight or obesity<\/b><\/h3>
STEP 2: obesity with type 2 diabetes<\/b><\/h3>
SELECT: cardiovascular outcomes in obesity without diabetes<\/b><\/h3>
Pharmacokinetic reviews<\/b><\/h3>
What Makes Semaglutide Different From Natural GLP-1?<\/b><\/h2>
What Makes Semaglutide Important in Current Research?<\/b><\/h2>
Research Considerations and Limitations<\/b><\/h2>
FAQ<\/b><\/h2>
Conclusion<\/b><\/h2>
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →