Selank: Anti-Anxiety Neuropeptide Research Guide
Written bySpartan Research Team
Selank is a synthetic heptapeptide developed by the Institute of Molecular Genetics of the Russian Academy of Sciences and the Institute of Pharmacology of the Russian Academy of Sciences in the early 2000s. It’s based on the immunomodulatory tetrapeptide tuftsin (Thr-Lys-Pro-Arg) with an additional tripeptide sequence added to extend its stability and central activity. Where most neuropeptides in the anxiolytic category work by sedating the CNS, Selank’s profile is different: it targets anxiety-relevant pathways without producing the sedation, tolerance, or physical dependence seen with benzodiazepine compounds in preclinical models. That combination has made it one of the more carefully studied neuropeptides in Russian and Eastern European pharmacological research.
- Seregin et al. documented Selank’s anxiolytic effects in elevated plus-maze models, with activity comparable to phenibut and diazepam but without motor impairment or sedation in treated animals (PMID 19916388).
- Selank has been shown to modulate GABA-A receptor subunit expression and stabilize GABAergic transmission without direct benzodiazepine binding site activity, suggesting an indirect GABAergic mechanism.
- Uchakina et al. (2008) demonstrated that Selank significantly suppressed IL-6 production in stimulated immune cell cultures, pointing to an anti-inflammatory neuroimmune component in its mechanism (PMID 18683560).
What Is Selank?
The peptide’s full sequence is Thr-Lys-Pro-Arg-Pro-Gly-Pro. It was synthesized by adding the tripeptide Pro-Gly-Pro to the C-terminus of tuftsin, and that addition dramatically changed its pharmacokinetics. Tuftsin itself degrades rapidly in biological systems. Selank is considerably more stable, with studies suggesting meaningful CNS penetration following intranasal administration in animal models. That stability matters for research purposes: a compound that’s gone within minutes is harder to study than one that maintains activity long enough to produce measurable behavioral and biochemical outcomes.
Tuftsin has well-documented immunomodulatory properties, and Selank carries some of that biological heritage. But the anxiety research focus emerged from behavioral pharmacology studies in the early 2000s that showed the compound’s effects on GABA-related signaling and stress-axis hormones. Selank became particularly interesting to researchers studying non-sedating anxiolytic mechanisms because it didn’t fit the usual receptor-binding model for that class of compounds.
Selank is available from Spartan Peptides for laboratory and in vitro research use. View product details.
GABA Modulation: What the Research Shows
One of the central debates in Selank research is exactly how it produces its anxiolytic-like behavioral effects. It doesn’t bind directly to GABA-A receptor benzodiazepine sites in the way that classic anxiolytic compounds do. What’s been found instead is more indirect: Selank administration in animal models appears to alter the expression of GABA-A receptor subunits in the hippocampus and cortex, and it stabilizes tonic GABAergic transmission without the receptor downregulation that characterizes benzodiazepine tolerance.
The Seregin research group’s work at the Institute of Pharmacology produced a series of papers examining these effects across different behavioral anxiety paradigms. Elevated plus-maze testing, open-field behavior, and conflict-test models all showed consistent anxiolytic-like outcomes. What didn’t show up: motor impairment, sedation, or amnesia at doses producing anxiolytic effects. That’s the pharmacological separation that distinguishes Selank’s profile from classic GABAergic drugs in preclinical data.
The enkephalin connection is worth mentioning. Some Russian research groups proposed that Selank’s activity involves partial enkephalinase inhibition, which would slow the degradation of endogenous enkephalins and prolong their effects on GABA interneurons. This mechanism, if confirmed, would place Selank in a different mechanistic category than direct GABA modulators and could explain some of the observed differences in tolerance profile.
IL-6 Regulation and Neuroimmune Interactions
Selank’s tuftsin heritage shows up clearly in its immunological activity. Tuftsin is a fragment of IgG that stimulates macrophage and neutrophil activity, and Selank carries similar immunomodulatory properties. The more interesting finding for neuropharmacology researchers is its effect on cytokine profiles, particularly IL-6.
Uchakina and colleagues published data in 2008 (PMID 18683560) showing that Selank significantly reduced IL-6 production in human peripheral blood mononuclear cells stimulated with phytohemagglutinin. The reduction was dose-dependent and selective: other cytokines tested showed less pronounced changes. Why does that matter for anxiety research? IL-6 is a pro-inflammatory cytokine that’s been associated with hypothalamic-pituitary-adrenal (HPA) axis activation and corticotropin-releasing hormone expression. The idea that an anxiolytic compound might also suppress neuroimmune inflammation adds a potentially independent mechanism to Selank’s behavioral effects.
This dual profile, anxiolytic behavior plus immunomodulatory activity, is actually consistent with Selank’s tuftsin origins. Researchers studying the intersection of stress physiology and immune function have found it a useful tool precisely because it seems to touch both systems.
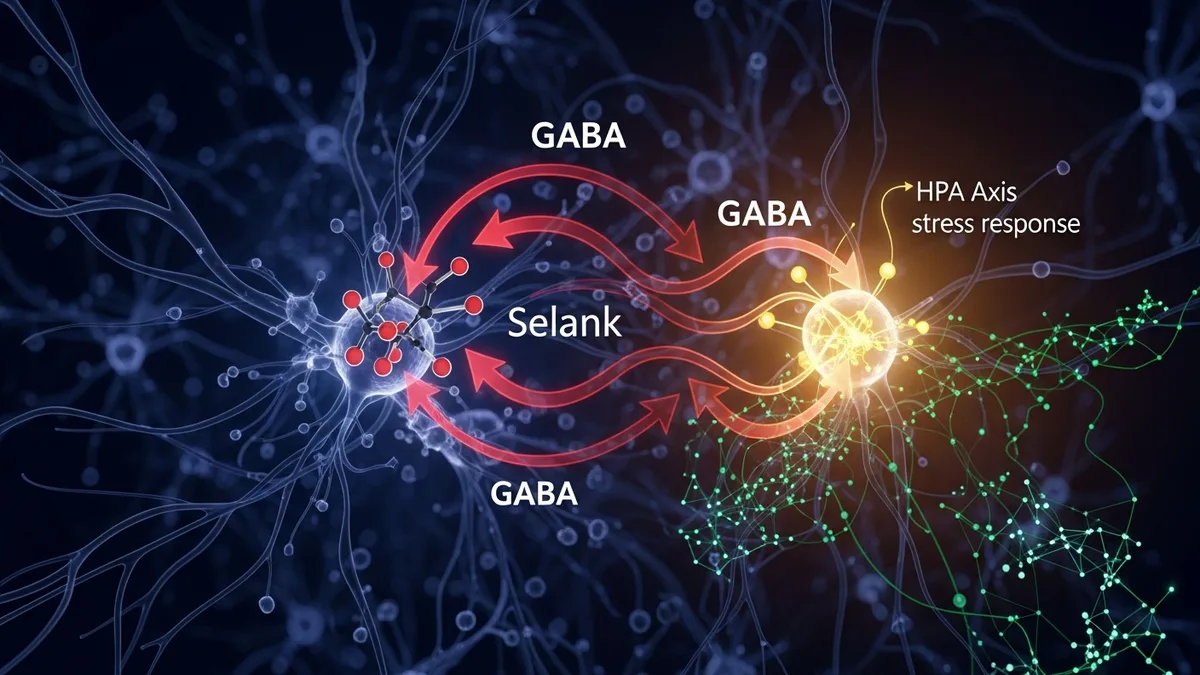
Selank modulates GABA-A receptor activity and blunts HPA axis stress responses, producing anxiolytic effects in rodent behavioral models. Explore Spartan Peptides catalog.
BDNF and Cognitive Effects in Preclinical Models
Brain-derived neurotrophic factor (BDNF) has attracted attention in Selank research. Several studies from Russian groups reported that Selank administration in rodents produced elevated hippocampal BDNF expression, and that this effect was observed at doses similar to those producing anxiolytic behavioral outcomes. BDNF plays key roles in synaptic plasticity, long-term potentiation, and learning and memory consolidation, which is why its regulation by a putative anxiolytic compound raises broader questions about the compound’s CNS effects.
Behavioral data in these models showed improvements in passive and active avoidance tasks alongside the BDNF changes, but the causal relationship remains unclear. It’s not established from the available data whether BDNF upregulation drives the cognitive outcomes, accompanies them, or is an epiphenomenon. That’s an honest summary of where the field is. The data are suggestive, not definitive, and most researchers in this space would agree more mechanistic work is needed.
Seregin Group Research: The Core Evidence Base
The most systematic Selank research has come from the Seregin group and their collaborators at the Institute of Molecular Genetics. Their published work spans multiple behavioral paradigms and covers dose-response characterization, mechanism studies, and comparisons with reference anxiolytic compounds. Seregin et al. (2009, PMID 19916388) directly compared Selank to phenibut and diazepam in elevated plus-maze models in Wistar rats. Selank produced anxiolytic-like effects at 300 micrograms per kilogram (intranasal), comparable in magnitude to diazepam at 1 milligram per kilogram (intraperitoneal), but without the motor coordination impairment measured on rotarod testing.
That selectivity index, anxiolytic efficacy without motor impairment, is the metric that’s made Selank worth studying as a pharmacological tool. For researchers designing experiments where anxiety-related behaviors need modulation without confounding the motor performance data, compounds with this separation are genuinely useful.
The group also published on chronic administration, finding no evidence of tolerance development over extended dosing periods in rodents, and no rebound anxiety after discontinuation. Those findings are in the published literature, though replication by independent groups outside Russia has been limited.
Intranasal Delivery and CNS Penetration
One of the more practically relevant aspects of Selank research involves delivery route. Most of the behavioral and biochemical data from Russian studies used intranasal administration, and there’s a good mechanistic reason for that. Intranasal delivery of neuropeptides can exploit olfactory and trigeminal nerve pathways to bypass the blood-brain barrier, delivering compounds directly to CNS tissue. For a heptapeptide that wouldn’t be expected to have high BBB permeability by standard pharmacokinetic rules, this delivery route effectively sidesteps the penetration problem.
Intranasal peptide pharmacokinetics are genuinely complex, and absorption efficiency varies significantly with formulation, volume, and species. The rat nasal anatomy differs from human anatomy in ways that affect translational relevance. Researchers working with Selank should be aware that in vitro characterization of the compound doesn’t directly translate to the in vivo delivery dynamics seen in rodent behavioral studies.
Research Considerations and Sourcing
For laboratory applications, Selank is typically used as a reference compound in anxiety, stress biology, and neuroimmunology research. Its distinct profile relative to benzodiazepines makes it useful in studies where GABAergic mechanism comparison is part of the experimental design.
- Purity verification: Research-grade Selank should carry HPLC documentation. Spartan Peptides supplies Selank at verified purity for laboratory use.
- Storage: Lyophilized peptides require cold storage. Reconstitute with bacteriostatic water for in vitro work. See the reconstitution guide for protocol details.
- Dose and delivery context: Published protocols used intranasal routes in rodent models. In vitro studies use concentration ranges derived from the in vivo data as a starting reference.
Related research tools in the neuropeptide space include Semax (another Russian-developed neuropeptide with BDNF-related activity) and Selank itself, both available from Spartan Peptides for in vitro and preclinical research use.
Frequently Asked Questions
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →