Cognitive Peptides for Brain Fog 2026: Semax, NAD+, and Pinealon Research Guide
Written bySpartan Research Team
Brain fog shows up in research literature as a cluster of measurable cognitive deficits: slowed processing speed, reduced working memory capacity, and impaired executive function. The cognitive performance research community has narrowed its focus to three compounds with the strongest preclinical data in these specific areas: Semax, NAD+, and Pinealon. Nootropic-focused biohackers studying cognitive restoration and neuroprotection pathways consistently cite this stack as the most mechanism-grounded starting point in the peptide space. This guide covers what each one actually does and why the research community finds the combination compelling.

Semax: The BDNF Upregulator That the Nootropic Research Community Has Rallied Around
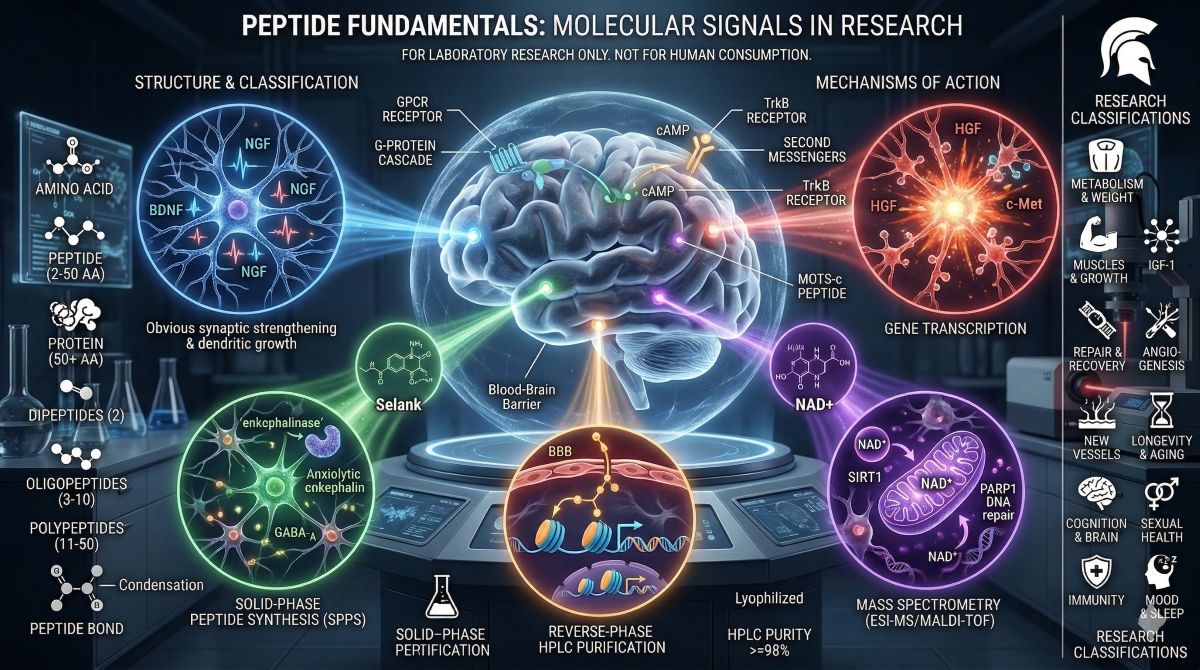
Semax is a synthetic ACTH(4-7)PGP analog — meaning it is derived from a fragment of adrenocorticotropic hormone with a Pro-Gly-Pro extension that dramatically extends its biological half-life and enables CNS penetration. Biohackers and cognitive performance researchers who have moved past lion’s mane and racetams consistently land on Semax because its mechanism is unusually well-defined for the nootropic space: it upregulates brain-derived neurotrophic factor (BDNF) expression in the cortex and hippocampus.
BDNF is the protein most directly responsible for neuroplasticity — the brain’s ability to form new synaptic connections, strengthen active circuits, and maintain structural integrity under cognitive load or stress. The research community studying brain fog mechanisms has focused heavily on BDNF because low BDNF expression consistently correlates with the cognitive signature that brain fog describes: slow processing, poor working memory retrieval, difficulty sustaining focused attention. Semax’s documented ability to raise BDNF levels in neural tissue is what makes it mechanistically relevant to this symptom cluster.
Preclinical research published in European Journal of Pharmacology (Inozemtseva et al., 2024) documented significant antidepressant-like and antistress effects of Semax in male rats exposed to chronic unpredictable stress models, with BDNF/NF-kB signaling implicated as the primary mechanism (PMID: 39442746). Rodent studies using the Morris Water Maze and novel object recognition tasks have found dose-dependent improvements in spatial memory and learning acquisition in Semax-treated subjects — the same cognitive domains that brain fog most visibly impairs in human self-reports.
The nootropic research community has also studied Semax for its attentional effects. Research dosing frameworks in animal models range from 25 mcg/kg to 150 mcg/kg. Intranasal delivery is the most studied route for CNS-targeted research given direct nasal-to-brain transport via the olfactory epithelium — a delivery pathway that the cognitive performance research community finds particularly relevant for compounds targeting cortical function. Researchers studying BDNF-dependent neuroplasticity, cognitive resilience under chronic stress, or working memory mechanisms consistently cite Semax as the most mechanistically well-characterized nootropic peptide in the current research toolkit.
Research Compound: Semax (ACTH(4-7)PGP) — Spartan Peptides Research Grade
NAD+: The Mitochondrial Fix That Biohackers Studying Cognitive Decline Keep Coming Back To
The cognitive performance research community has increasingly converged on NAD+ as the foundational substrate that other nootropic compounds depend on. The reason is straightforward: neurons are the highest-energy cells in the body, and NAD+ (nicotinamide adenine dinucleotide) is the central electron carrier that mitochondria require to produce ATP. When NAD+ availability drops, mitochondrial output drops — and in neural tissue, that shows up cognitively before it shows up anywhere else.
Research has documented that brain NAD+ concentration declines approximately 30-50% between early adulthood and senescence in rodent models. Biohackers studying cognitive aging and the brain fog that accompanies it have cited this NAD+ decline as the upstream variable that compounds like Semax work within — BDNF-mediated neuroplasticity requires ATP to execute, and depleted mitochondrial substrate capacity creates a ceiling on what any upstream nootropic mechanism can accomplish. Optimization-focused researchers studying cognitive restoration have described this as the “mitochondrial floor problem”: you cannot fully optimize neuroplasticity with Semax or other BDNF modulators if the energy substrate availability is compromised.
Research published in Brain (Lagartos-Donate et al., 2026) demonstrated that NAD administration ameliorated loss of the REST neuroprotective factor in Alzheimer’s disease pathology models — REST is a transcription factor that suppresses genes associated with neuronal apoptosis, and its decline is one of the documented early markers of cognitive deterioration (PMID: 41709697). Separately, research in Neuromolecular Medicine (Alameen et al., 2026) examined SIRT1 activators as geroprotective agents in brain aging — SIRT1 is an NAD+-dependent enzyme, and its role in neuroprotection ties directly into the NAD+ availability picture (PMID: 41934491).
For nootropic-focused biohackers studying the full NAD+ mechanism — cellular energy production, DNA repair via PARP enzymes, sirtuin activation, and neuroprotective gene regulation — the complete NAD+ research guide covers the mechanistic detail, dosing frameworks for preclinical models, and the longevity research intersection.
Pinealon: The Epigenetic Neuroprotector That the Cognitive Aging Research Community Uses
Pinealon is the research name for the EDR tripeptide (Glu-Asp-Arg) — a short-chain peptide bioregulator originally isolated from pineal gland tissue through the work of Khavinson and colleagues in Russian gerontology research. Among cognitive performance researchers who track neuroprotection alongside acute cognitive enhancement, Pinealon occupies a distinct position: it is not primarily a “sharpen focus now” compound but a structural neuroprotector — something studied for its role in maintaining the neural architecture that cognitive performance depends on over time.
The mechanism proposed for Pinealon is epigenetic: the EDR tripeptide is theorized to penetrate cell membranes and directly modulate transcriptional activity of genes involved in neuroprotection, mitochondrial integrity, and apoptosis resistance in neural tissue. This is mechanistically different from Semax (receptor-mediated BDNF signaling) and NAD+ (substrate-level mitochondrial support). Pinealon is studied as a gene-level regulator in neurons — an upstream neuroprotective influence that may determine whether the neural circuitry Semax and NAD+ are optimizing stays structurally intact under aging or chronic oxidative stress.
Preclinical research in aged rodent models has documented improvements in spatial memory task performance and measurable reductions in hippocampal neuronal apoptosis markers in Pinealon-treated subjects compared to vehicle controls. The cognitive aging research community has found these results particularly relevant given that hippocampal neuronal loss is a direct driver of the working memory and orientation deficits associated with both age-related cognitive decline and the chronic brain fog that motivates much of the nootropic research community’s compound exploration.
Researchers combining Pinealon with NAD+ in aging model protocols have reported synergistic effects on mitochondrial integrity and apoptosis resistance in hippocampal tissue — consistent with the hypothesis that epigenetic neuroprotection and mitochondrial energy substrate availability address the same underlying neural deterioration from complementary angles.
Research Compound: Pinealon (EDR Tripeptide) — Spartan Peptides Research Grade
🔬 Key Research Findings
- Semax (ACTH(4-7)PGP) consistently upregulates BDNF in cortical and hippocampal tissue in preclinical models, with dose-dependent improvements in working memory, spatial learning acquisition, and cognitive resilience under chronic stress — the same variables the cognitive performance research community tracks as brain fog markers.
- Brain NAD+ declines ~30-50% with age in rodent models, correlating with measurable cognitive task deficits; preclinical NAD+ restoration protocols partially reverse these deficits and preserve neuroprotective gene expression including REST, establishing NAD+ as the mitochondrial foundation that upstream nootropic mechanisms depend on.
- Pinealon (EDR tripeptide) in aged rodent research is associated with improved spatial memory performance and reduced hippocampal apoptosis markers — a structural neuroprotective profile that complements the acute cognitive enhancement mechanisms of Semax and the energy substrate role of NAD+.
- Researchers combining Semax and NAD+ in preclinical cognitive models find the pairing mechanistically additive: NAD+ addresses mitochondrial energy substrate availability while Semax provides BDNF-mediated upstream neuroplasticity signaling — complementary mechanisms rather than redundant ones.
Compound Comparison: Where Each One Fits in a Cognitive Research Stack
| Compound | Mechanism | Research Focus | Format | Product |
|---|---|---|---|---|
| Semax | BDNF upregulation via ACTH analog / neuropeptide signaling | Working memory, neuroplasticity, attentional performance, cognitive stress resilience | Lyophilized peptide (intranasal / SC in research models) | Semax |
| NAD+ | Mitochondrial electron carrier; SIRT1 and PARP substrate; DNA repair cofactor | Cognitive aging, neuronal energy metabolism, neuroprotective gene expression (REST, SIRT1) | Lyophilized powder (IV / SC in research models) | NAD+ 750mg |
| Pinealon | EDR tripeptide epigenetic modulation in neural tissue | Neuroprotection, hippocampal neuronal preservation, cognitive aging in rodent models | Lyophilized tripeptide (SC in research models) | Pinealon |
How the Cognitive Research Community Has Combined These Compounds
Nootropic-focused biohackers and cognitive performance researchers have increasingly studied these three compounds in combination rather than isolation — the mechanistic rationale is that they address genuinely different upstream variables in the cognitive performance equation, making them complementary rather than redundant.
The framing that the cognitive performance research community uses: Semax addresses the neuroplasticity signal (BDNF-mediated synapse formation and maintenance), NAD+ addresses the energy substrate floor (mitochondrial ATP production capacity that all neural function depends on), and Pinealon addresses the structural neuroprotection layer (epigenetic maintenance of the neural architecture that the other two compounds are optimizing). Researchers studying brain fog mechanisms through this lens argue that addressing only one layer — say, BDNF signaling without mitochondrial support — produces incomplete results because the mechanisms are interdependent.
In preclinical aging models, multi-compound protocols using these agents have documented cognitive performance outcomes that exceed single-compound baselines. Pinealon protocols have been studied at 10-28 day durations in aged rodents, with endpoints focused on structural and apoptosis markers. Semax and NAD+ combinations have been examined in shorter-duration cognition-focused studies with behavioral task endpoints. None of this constitutes human use guidance. All research use should comply with applicable IRB and institutional protocols. For additional context on multi-compound nootropic research frameworks, see the Focus and Clarity research category.
Frequently Asked Questions
Why does the cognitive research community focus on Semax specifically for brain fog research?
Semax has an unusually well-defined mechanism for the nootropic peptide space: it upregulates BDNF expression in the cortex and hippocampus through its ACTH(4-7)PGP analog structure. BDNF directly governs neuroplasticity — synaptic formation, circuit strengthening, and structural maintenance under stress. The specific cognitive deficits that brain fog describes (slow processing, poor working memory, impaired attention) map directly to the neural domains where BDNF expression is most consequential. That mechanistic specificity, backed by preclinical research in stress models and memory tasks, is why Semax has become the reference nootropic peptide for brain fog research applications.
How does NAD+ fit into a cognitive peptide research stack?
NAD+ functions as the mitochondrial energy substrate that all neuroplasticity mechanisms ultimately depend on. Neurons are the highest-energy cells in the body; declining NAD+ availability with age directly impairs mitochondrial ATP production in neural tissue. Cognitive performance researchers studying brain fog increasingly describe NAD+ as the “floor” that other nootropic compounds operate within — BDNF-mediated neuroplasticity via Semax requires ATP to execute, and NAD+ determines whether that substrate is available. Research has also documented NAD+’s role in preserving neuroprotective gene expression (REST, SIRT1) that declines in cognitive aging models.
What is Pinealon and how is it different from Semax in cognitive research?
Pinealon (EDR tripeptide, Glu-Asp-Arg) is an epigenetic neuroprotector rather than an acute cognitive enhancer. Where Semax works at the receptor/signaling level to upregulate BDNF, Pinealon is proposed to enter cell nuclei and modulate transcriptional activity of genes governing neuroprotection and apoptosis resistance in neural tissue. The cognitive aging research community uses Pinealon for its structural neuroprotective profile — preserving hippocampal neuronal density and reducing apoptosis markers — rather than for immediate working memory or attention effects. It complements Semax by protecting the neural architecture that Semax-enhanced neuroplasticity depends on.
Can these three compounds be studied together in a cognitive research protocol?
Yes — preclinical research has examined multi-compound protocols combining Semax, NAD+, and Pinealon. Their mechanisms are non-overlapping: Semax provides BDNF-mediated neuroplasticity signaling, NAD+ provides mitochondrial energy substrate support, and Pinealon provides epigenetic neuroprotection at the gene-regulation level. The cognitive performance research community describes these as addressing three distinct layers of the brain fog mechanism simultaneously. All compound use should be for in vitro and animal research purposes only, in compliance with applicable IRB guidelines.
Research Disclaimer
For Research Use Only: All compounds discussed in this article are intended exclusively for laboratory and preclinical research applications. This content is provided for scientific and educational purposes only and does not constitute medical advice, treatment recommendations, or clinical guidance. These compounds are not approved for human consumption by the FDA or equivalent regulatory bodies. Researchers should conduct all work in compliance with applicable institutional review board (IRB) protocols, local regulations, and ethical guidelines. Spartan Peptides compounds are sold strictly for in vitro and research model applications. Not for human consumption.
References
- Inozemtseva LS et al. “Antidepressant-like and antistress effects of the ACTH(4-10) synthetic analogs Semax and Melanotan II on male rats in a model of chronic unpredictable stress.” Eur J Pharmacol. 2024 Dec;984:177068. PMID: 39442746
- Lagartos-Donate MJ et al. “Loss of REST associated with Alzheimer’s disease pathology is ameliorated by NAD.” Brain. 2026;149(4):1208-1223. PMID: 41709697
- Alameen AAM et al. “SIRT1 Activators as Geroprotective Agents in Brain Aging.” Neuromolecular Med. 2026. PMID: 41934491
Spartan Research Library
Go Deeper on the Science
Browse study indexes, compound comparisons, and protocol stacks in the Spartan Peptides Research Library.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →