Complete Guide to Nootropic and Cognitive Peptides
Written bySpartan Research Team
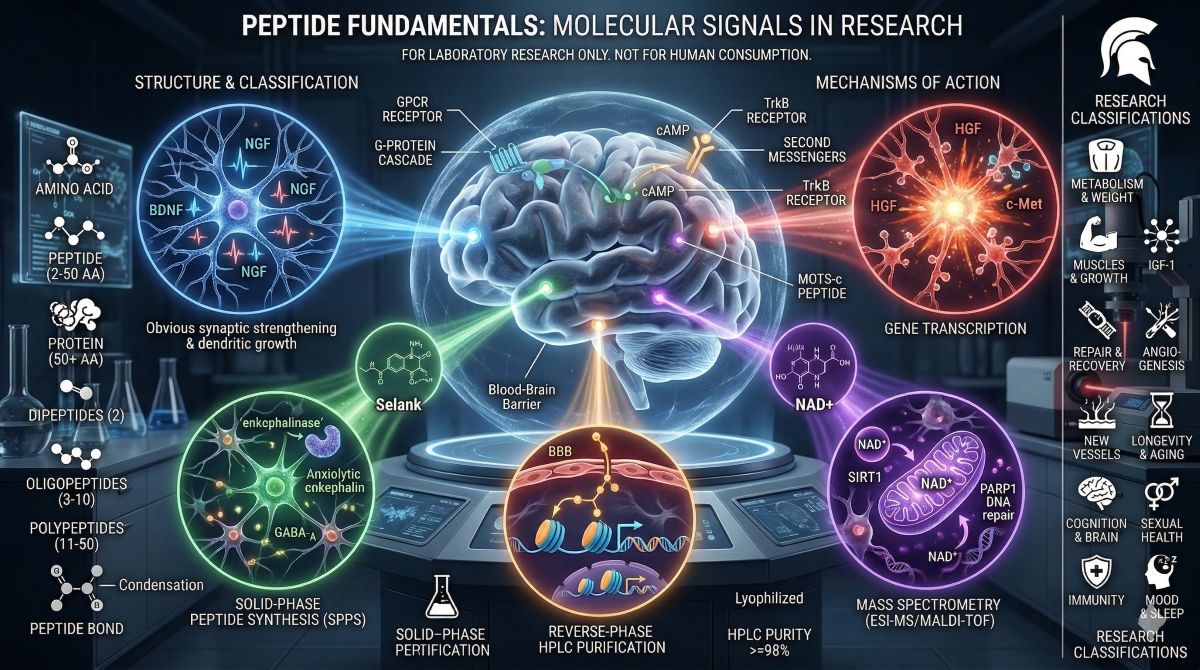
Cognitive peptides and nootropic research compounds occupy a distinct corner of the peptide science literature — one that has grown substantially as investigators mapped the molecular biology of memory consolidation, neuroplasticity, and neuroprotection with increasing precision. The question driving this research is mechanistic: which signaling pathways govern learning, attention, and neuronal resilience, and can targeted peptide compounds modulate those pathways in controlled research models?
This guide covers the primary cognitive peptides and brain peptides currently under scientific investigation. The compounds reviewed here — Semax, Selank, Pinealon, Dihexa, and NAD+ — represent five mechanistically distinct approaches to studying central nervous system biology. Semax acts through BDNF and NGF induction; Selank modulates GABAergic and enkephalinase pathways; Pinealon penetrates the blood-brain barrier to regulate neuroprotective gene expression; Dihexa acts downstream of HGF/Met signaling with documented synaptic efficacy orders of magnitude beyond reference compounds; NAD+ addresses the metabolic and genomic maintenance functions that sustain neuronal viability over time. Each mechanism is grounded in a distinct body of peer-reviewed research, and each addresses a different rate-limiting process in cognitive neurobiology.
All compounds discussed here are research peptides supplied strictly for laboratory investigation. They are not approved for human consumption and are referenced exclusively in the context of scientific research.
The Neurobiology of Cognition: What Cognitive Peptides Target
Before examining individual compounds, it is useful to map the biological processes that cognitive peptide research targets. Cognition — memory formation, attention, processing speed, executive function — emerges from the interaction of synaptic plasticity, neurotrophic factor signaling, neurotransmitter regulation, and neuronal metabolic integrity. Research peptides in this category intervene at different points in this system.
Brain-Derived Neurotrophic Factor (BDNF) and Neuroplasticity
BDNF is a member of the neurotrophin family that binds TrkB receptors on neurons to promote synaptic strengthening, dendritic branching, and long-term potentiation (LTP) — the cellular correlate of memory consolidation. BDNF levels are regulated by neuronal activity, stress, sleep, and various pharmacological agents. Reduced BDNF expression is associated with age-related cognitive decline, major depressive disorder, and neurodegenerative disease across multiple human studies. Research compounds that elevate BDNF expression or activate TrkB signaling are therefore studied as potential tools for investigating the molecular basis of neuroplasticity.
Nerve Growth Factor (NGF) and Cholinergic Neurons
NGF is the founding member of the neurotrophin family, acting through TrkA receptors on cholinergic neurons of the basal forebrain — the primary neuronal population that degenerates in Alzheimer’s disease and is associated with memory and attentional processing. NGF is produced by hippocampal and cortical neurons and transported retrogradely to support cholinergic neuron survival and function. Research compounds that increase NGF expression are studied for their capacity to maintain cholinergic neuron integrity in models of neurodegeneration and cognitive aging.
HGF/Met Signaling and Synaptogenesis
Hepatocyte growth factor (HGF) and its receptor Met (c-Met) were originally characterized in liver regeneration research but are now established regulators of neuronal migration, axon guidance, and synaptogenesis in the CNS. HGF/Met signaling promotes dendritic spine formation and synaptic density — structural correlates of learning capacity. Research into HGF/Met pathway modulation in the brain is an area of active investigation, particularly in the context of compounds that can cross the blood-brain barrier efficiently and produce meaningful synaptic effects at low concentrations.
GABAergic Modulation and Stress-Anxiety Biology
The gamma-aminobutyric acid (GABA) system is the primary inhibitory neurotransmitter network in the mammalian CNS, and its dysregulation is implicated in anxiety disorders, stress-induced cognitive impairment, and insomnia. Peptide compounds that modulate GABA-A receptor activity or regulate the metabolism of endogenous anxiety-reducing peptides (such as enkephalins) are studied for their effects on stress response biology and the anxiety-cognition interface — the well-documented phenomenon by which excessive HPA axis activation impairs hippocampal-dependent memory consolidation.
Neuronal Metabolism, NAD+, and Mitochondrial Function
The brain accounts for approximately 20% of the body’s total energy consumption at rest, and neurons are among the most metabolically active and mitochondria-dense cells in the body. NAD+-dependent processes — sirtuin-mediated gene regulation, PARP-mediated DNA repair, and mitochondrial electron transport chain function — are as relevant in neurons as in peripheral tissues, and the age-related decline in NAD+ that impairs metabolic function generally has specific consequences for neuronal health: reduced capacity for DNA repair in post-mitotic neurons that cannot dilute damage through division, impaired mitochondrial quality control, and reduced SIRT1 activity governing neuroprotective gene expression.
| ★ | Research context: The blood-brain barrier (BBB) is the primary pharmacokinetic constraint on CNS-active compounds. Peptides larger than approximately 500–600 Da are generally excluded by the BBB under normal conditions, though specific transport mechanisms exist for some peptides. Several cognitive research compounds discussed here have been specifically designed or selected for BBB penetration — a pharmacokinetic property that must be verified experimentally for each compound rather than assumed. |
Cognitive and Nootropic Peptides: Compound Profiles
- Semax
Semax is a synthetic heptapeptide (Met-Glu-His-Phe-Pro-Gly-Pro) derived from the ACTH(4-7) sequence, with a Pro-Gly-Pro extension at the C-terminus that confers resistance to enzymatic degradation and extends functional half-life. It was developed at the Institute of Molecular Genetics of the Russian Academy of Sciences and has been the subject of over three decades of Russian and Eastern European neuroscience research. Semax is administered intranasally in the majority of research models, with nasal mucosa providing a partially direct route to the olfactory bulb and CNS that bypasses some of the BBB restriction applied to systemic administration. Detailed mechanistic and study data are available in our Semax: Enhancing Mental Performance research overview.
Semax Mechanism: BDNF, NGF, and ACTH Receptor Partial Agonism
Semax’s primary documented mechanisms are induction of BDNF and NGF expression in the hippocampus and frontal cortex, partial agonism at melanocortin receptors (particularly MC4R), and modulatory effects on dopaminergic and serotonergic neurotransmission. The BDNF-inducing effect is the most thoroughly characterized: rat models administered Semax intranasally show dose-dependent increases in hippocampal BDNF mRNA expression within hours, with corresponding downstream activation of TrkB and downstream CREB phosphorylation — the transcription factor that mediates BDNF’s effects on synaptic plasticity and long-term potentiation.
The NGF induction effect is particularly relevant for cholinergic neuron research. In models of focal ischemia, Semax administration produced increases in NGF expression in the ischemic penumbra alongside reduced neuronal apoptosis and improved functional recovery metrics (Dmitrieva et al., 2010). The mechanism linking Semax administration to NGF gene activation is not fully characterized, but is thought to involve ACTH receptor-mediated cAMP elevation and PKA-dependent transcription factor activation at NGF promoter sites.
Semax also modulates the serotonergic system: studies in rats have documented increased tryptophan hydroxylase expression and elevated serotonin turnover in the prefrontal cortex following Semax administration, which is consistent with its documented effects on attention and stress response in animal behavioral paradigms.
Spartan Peptides supplies Semax research peptide for laboratory investigation. For a combined research overview of Semax alongside Selank in the stress-cognition context, see Semax vs. Selank.
- Selank
Selank is a synthetic heptapeptide (Thr-Lys-Pro-Arg-Pro-Gly-Pro) derived from the endogenous immunomodulatory peptide tuftsin (Thr-Lys-Pro-Arg), with the same C-terminal Pro-Gly-Pro extension used in Semax to stabilize the molecule against peptidase activity. It was also developed at the Institute of Molecular Genetics in Moscow, alongside Semax, and has been studied primarily for its anxiolytic and cognition-modulating properties in preclinical and limited human research.
Selank Mechanism: Enkephalinase Inhibition and GABAergic Modulation
Selank’s most thoroughly characterized mechanism is inhibition of enkephalinase — the enzyme responsible for degrading endogenous enkephalins (Met-enkephalin, Leu-enkephalin), which are opioid peptides with anxiolytic and mood-stabilizing properties. By slowing enkephalin degradation, Selank prolongs the activity of these endogenous anxiolytic signals without directly activating opioid receptors — a selectivity distinction with research relevance for separating anxiety reduction from opioid receptor-mediated effects.
Selank also shows evidence of GABA-A receptor potentiation. In rat electrophysiology studies, Selank administration produced changes in GABA-A receptor subunit expression consistent with anxiolytic benzodiazepine-like effects, but without the receptor downregulation associated with chronic benzodiazepine exposure. The specific GABA-A subunit interaction has not been fully characterized, which limits mechanistic comparison with established GABA-A modulators.
In behavioral research paradigms, Selank has produced consistent anxiolytic effects in rodent models (elevated plus maze, open field test) without the motor impairment or sedation associated with benzodiazepine reference compounds. A small human trial by Semenova et al. (2010) found that Selank administration in patients with generalized anxiety disorder produced anxiolytic effects comparable to medazepam, a reference benzodiazepine, with a different side effect profile. The absence of larger controlled human trials limits the translational interpretation of this finding.
A detailed mechanistic and study comparison of Selank alongside other anxiety/cognitive peptides is available in our Selank: Anxiety, Stress, and Nootropic Peptide Research.
- Pinealon
Pinealon is a tripeptide (Glu-Asp-Arg) developed by Vladimir Khavinson’s group at the St. Petersburg Institute of Bioregulation and Gerontology — the same laboratory responsible for Epithalon. At three amino acids and a molecular weight of approximately 403 Da, Pinealon is among the smallest research peptides studied for CNS effects, and its size is mechanistically relevant: the short chain length gives it favorable properties for crossing biological barriers, including the BBB. Research has documented Pinealon’s capacity to protect neurons from oxidative stress, modulate pineal gland gene expression, and attenuate age-related cognitive decline in animal models. A research overview is available in our Supporting Cognitive Functions with Pinealon.
Pinealon Mechanism: Neuroprotective Gene Regulation and Epigenetic Activity
Pinealon’s documented mechanism centers on the regulation of gene expression in neural tissue, particularly genes involved in antioxidant defense, circadian rhythm regulation, and neuronal survival. At the cellular level, it has been shown in in vitro models to reduce oxidative stress-induced neuronal apoptosis by modulating the expression of Bcl-2 family proteins — shifting the balance toward anti-apoptotic signaling — and by reducing reactive oxygen species accumulation in neuronal mitochondria.
The epigenetic dimension of Pinealon’s activity has been documented by Khavinson et al. in studies showing that short peptides of this class can interact directly with gene promoter regions and histone-DNA complexes, acting as gene expression regulators through chromatin remodeling rather than classical receptor binding. This mechanism, if confirmed by independent groups, would represent a distinct class of peptide action — one that operates at the level of transcriptional regulation rather than receptor pharmacology.
In aged rat models, Pinealon administration was associated with improved performance in Morris water maze tests (a standard hippocampal-dependent spatial memory paradigm) and reduced markers of oxidative stress in hippocampal tissue. The compound has also shown activity in models of retinal neurodegeneration — a finding consistent with its pineal origin and potential circadian biology effects, since retinal ganglion cells share embryological origin with pineal cells.
Spartan Peptides supplies Pinealon research compound. For combined Pinealon/Semax research in stress and sleep contexts, see Pinealon, Semax: Stress, Sleep, and Cognitive Research.
- Dihexa
Dihexa (N-hexanoic-Tyr-Ile-(6) aminohexanoic amide) is a synthetic peptidomimetic derived from angiotensin IV, designed by Joseph Harding and colleagues at Washington State University to resist peptidase degradation while retaining the HGF/Met signaling activity of its parent compound. It has attracted attention in the nootropic peptide research field for two reasons: its documented BBB penetrance by oral and systemic routes in rodent models, and its reported potency in cognitive performance assays at concentrations orders of magnitude below those required by reference nootropic compounds. A dedicated research overview is at our Dihexa: Nootropic Peptide and Cognitive Enhancement Research.
Dihexa Mechanism: HGF/Met Signaling, Synaptic Density, and Spinogenesis
Dihexa binds hepatocyte growth factor (HGF) and facilitates its interaction with the Met receptor (c-Met) on neurons — a receptor tyrosine kinase that, when activated in CNS tissue, promotes dendritic spine formation (spinogenesis), synaptic density increase, and axonal outgrowth. HGF/Met signaling in the brain is distinct from peripheral HGF biology: in the CNS, it operates as a neurotrophic and synaptogenic factor rather than a mitogen.
The published potency data from Harding’s laboratory are striking. In a 2013 paper (Benoist et al., 2013, Journal of Pharmacology and Experimental Therapeutics), Dihexa outperformed BDNF in spatial learning assays in aged rats by a factor estimated at seven orders of magnitude in apparent molar potency — 10 million-fold more potent than BDNF when compared on a molar concentration basis in the models tested. This figure has attracted both research interest and methodological scrutiny: the comparison is across different administration routes and outcome measures, and independent replication of the full potency characterization has been limited. The spinogenesis data — showing increased dendritic spine density in cortical neurons following Dihexa treatment — provides a structural correlate for the behavioral findings, and spine density is directly related to synaptic connectivity and learning capacity.
Dihexa’s peptidomimetic design addresses a key limitation of conventional peptides: it is orally bioavailable in rodent models (a property rare in peptides of similar size), and it has a documented half-life substantially longer than natural angiotensin-derived peptides due to its engineered resistance to angiotensin-converting enzyme and aminopeptidases. These pharmacokinetic properties make it a tractable compound for in vivo CNS research in ways that many natural neuropeptides are not.
- NAD+ in Neurological Research
NAD+ is covered in depth in the Complete Guide to Anti-Aging Peptides for its systemic metabolic and DNA repair functions, but its CNS-specific research applications warrant dedicated attention here. Neurons are among the most NAD+-dependent cells in the body: they are post-mitotic (meaning they cannot dilute DNA damage through division), they maintain extremely high metabolic activity, and they express SIRT1 and SIRT3 at high levels relative to most peripheral tissues. The research implications of NAD+ depletion in neurons are therefore disproportionate to its systemic effects. The foundational NAD+ neuroscience data are reviewed in our NAD+ 750mg Complete Research Guide.
NAD+ Mechanism in Neurons: SIRT1, PARP1, and Neuroprotection
SIRT1 in neurons deacetylates and activates PGC-1alpha (promoting mitochondrial biogenesis), deacetylates p53 (reducing apoptotic signaling under genotoxic stress), deacetylates NF-kB RelA/p65 (attenuating inflammatory gene expression), and promotes the expression of neuroprotective factors, including BDNF. The NAD+/SIRT1 axis, therefore, connects cellular energy status directly to neuronal survival and synaptic plasticity gene expression — a mechanistic link that positions NAD+ repletion as a potential tool for studying the relationship between neuronal metabolic health and cognitive function.
PARP1-mediated NAD+ consumption in neurons deserves particular attention. Neurons experience oxidative DNA damage at rates proportional to their high metabolic activity and ROS production. PARP1 is activated by single-strand breaks and repairs them by consuming NAD+ to synthesize poly(ADP-ribose) (PAR) chains on histones, recruiting DNA repair enzymes. In aged neurons, elevated baseline DNA damage drives chronic PARP1 activation, accelerating NAD+ depletion and creating a feedback cycle: more damage → more PARP1 activity → less NAD+ → less SIRT1 activity → less mitochondrial quality control → more ROS → more damage.
NAD+ repletion research in neurological models has documented protection against PARP1-mediated NAD+ depletion following oxidative insults, preservation of mitochondrial membrane potential in aging neurons, and SIRT1-mediated attenuation of neuroinflammatory signaling in microglia. Whether these mechanistic findings translate to measurable cognitive outcomes in controlled research models is an active area of investigation.
Spartan Peptides supplies NAD+ research compound at ≥98% HPLC purity. For research into multi-compound protocols combining NAD+ with Semax and CJC-1295, see Energizer Bunny: NAD+, Semax, and CJC-1295 Stack Research.
Comparative Overview: Cognitive Research Peptides at a Glance
| Compound | Primary Mechanism | Key Research Finding | CNS Target | Class |
|---|---|---|---|---|
| Semax | BDNF/NGF induction; MC4R partial agonism; serotonin modulation | Hippocampal BDNF upregulation; neuroprotection in focal ischemia models (Dmitrieva et al., 2010) | TrkB/TrkA; hippocampus and frontal cortex | ACTH(4-7) analogue |
| Selank | Enkephalinase inhibition; GABA-A receptor modulation | Anxiolytic effects comparable to benzodiazepine in a small human trial (Semenov et al., 2010) | GABA-A; opioid peptide system | Tuftsin analogue |
| Pinealon | Neuroprotective gene regulation; anti-apoptotic signaling; epigenetic activation | Improved spatial memory in aged models; reduced hippocampal oxidative stress | Bcl-2 pathway; pineal/retinal neural tissue | Short regulatory tripeptide |
| Dihexa | HGF/Met receptor potentiation; dendritic spinogenesis | 7-log-unit potency advantage over BDNF in spatial learning paradigms (Benoist et al., 2013) | c-Met/HGF; synaptic density regulation | Angiotensin IV peptidomimetic |
| NAD+ | SIRT1/PARP1 axis; mitochondrial quality control; neuroinflammation modulation | PARP1-mediated neuroprotection; SIRT1-driven BDNF expression; mitochondrial preservation in aging neurons | SIRT1; PARP1; neuronal mitochondria | Coenzyme/metabolite |
Neuroprotection vs. Cognitive Enhancement: Two Research Frameworks

The cognitive peptide literature spans two distinct research frameworks that are often conflated but operate through different mechanisms and produce different experimental designs.
Neuroprotection Research
Neuroprotection research examines whether compounds can maintain or restore neuronal viability under conditions of pathological stress — ischemia, excitotoxicity, oxidative insult, or neurodegenerative disease progression. The endpoint is cell survival and functional recovery, measured against a control condition of defined injury. Semax’s most controlled data come from ischemia models; Pinealon’s most consistent findings are in aging and oxidative stress models; NAD+’s neuroprotective work centers on PARP1-driven cell death models. In these contexts, the research compounds are studied as tools for understanding cellular survival mechanisms, not cognitive enhancement per se.
Cognitive Enhancement Research
Cognitive enhancement research uses behavioral paradigms — Morris water maze, radial arm maze, novel object recognition, and fear conditioning — to measure memory acquisition, retention, and retrieval in animal models with defined cognitive deficits (aging, surgical lesion, and pharmacological impairment). Dihexa’s published data fall predominantly in this category: the spatial learning paradigm is a well-validated measure of hippocampal-dependent memory, and the head-to-head comparison with BDNF provides a reference point for potency assessment. Semax’s behavioral data — improved performance in memory tasks following hippocampal BDNF induction — spans both frameworks.
The distinction matters for research design because neuroprotection studies require a defined injury model, whereas cognitive enhancement studies can be conducted in healthy or aged animals without induced pathology. Most cognitive peptide research uses at least one of these frameworks, and the translational interpretation of findings differs substantially between them: a compound that protects neurons from ischemic injury is not necessarily the same as one that improves cognitive performance in a healthy aged brain, even if both are described as ‘neuroprotective’ in the literature.
| ★ | Methodological note: Behavioral neuroscience paradigms for cognitive research are well-validated within species but have imperfect translational reliability across species. Hippocampal-dependent spatial memory in rodents (Morris water maze) correlates with declarative memory systems in primates, but the correspondence is not one-to-one. Compounds producing strong effects in rodent cognitive paradigms have historically shown variable results when tested in humans, which is a standard caveat for preclinical cognitive neuroscience research. |
Research Evidence: Synthesis by Compound
Semax: Three Decades of Russian Neuroscience Data
Semax has the largest published evidence base of any compound in this group, reflecting its development as a pharmaceutical compound in Russia (where it is registered for medical use under conditions analogous to a clinical drug authorization). The Russian literature includes controlled human studies in ischemic stroke, attention deficit models, and cognitive aging, but these have not been replicated in Western clinical trials meeting contemporary evidence standards. The most methodologically rigorous independent data come from controlled animal studies documenting BDNF and NGF induction with statistical significance and dose-response characterization.
Dmitrieva et al. (2010) represent one of the strongest individual studies, documenting NGF elevation and neuroprotection in a rat focal ischemia model with well-defined outcome measures. Subsequent work from multiple Russian groups has replicated BDNF induction findings in hippocampal preparations using standardized assay methods. The consistency of these mechanistic findings across independent laboratories provides reasonable confidence in the BDNF/NGF mechanism, even where translational clinical data remain limited.
Selank: Anxiety-Cognition Interface Data
Selank’s evidence base is smaller than Semax’s but methodologically sound at the preclinical level. The enkephalinase inhibition mechanism has been confirmed by multiple groups using peptidase activity assays, and the behavioral anxiolytic effects in rodent paradigms are consistent across the published literature. The 2010 Semenova trial in generalized anxiety disorder patients — while small and conducted under Russian research protocols — provides human-context data for the anxiolytic effect and a reference point for comparison with benzodiazepine activity.
The cognitive dimension of Selank’s research is primarily indirect: by attenuating stress-induced HPA axis activation, which produces cortisol-mediated hippocampal damage under chronic conditions, the anxiolytic effect has the theoretical downstream consequence of protecting hippocampal integrity. This anxiety-cognition connection is well-established in the basic neuroscience literature independently of Selank-specific data.
Pinealon: Khavinson Group Data and Independent Verification Questions
Pinealon’s published evidence comes almost entirely from Khavinson’s St. Petersburg laboratory, the same group responsible for Epithalon research. The consistency of positive findings from a single research group is a legitimate methodological consideration, though it does not invalidate the findings — it simply makes independent replication a particularly important next step for the field. The mechanistic work on epigenetic peptide activity — short peptides interacting with chromatin — has been published in peer-reviewed journals and offers a theoretically coherent mechanism for observed gene expression effects.
The Morris water maze data in aged rats are consistent with the proposed neuroprotective mechanism, and the oxidative stress findings in hippocampal tissue provide a plausible mechanistic link between the compound’s gene regulatory effects and its behavioral outcomes. What the published literature does not yet contain is a controlled human trial, pharmacokinetic characterization in primates, or independent laboratory replication of the core behavioral findings.
Dihexa: High-Impact Data, Limited Independent Replication
Dihexa’s evidence base is anchored by Benoist et al. (2013) — a methodologically careful paper from an established laboratory published in a peer-reviewed pharmacology journal. The spatial learning data, the spine density histology, and the HGF/Met binding characterization are all presented with appropriate controls and statistical rigor. The extraordinary potency claim (seven orders of magnitude relative to BDNF) is striking but has a plausible mechanistic basis: Dihexa is not competing directly with BDNF at TrkB but acting on a different receptor system (Met), and the comparison is therefore not a direct potency comparison within the same assay.
The limitation of Dihexa’s evidence base is sparse independent replication. The Harding laboratory at Washington State has produced the foundational work, but the field has not yet produced the convergent findings from multiple independent groups that would be expected for a compound with such dramatic reported effects. This is not a disqualifying finding — many important compounds have narrow original evidence bases — but it is the appropriate scientific context for evaluating the current state of Dihexa research.
Combination Research: Cognitive Peptides and Multi-Compound Protocols
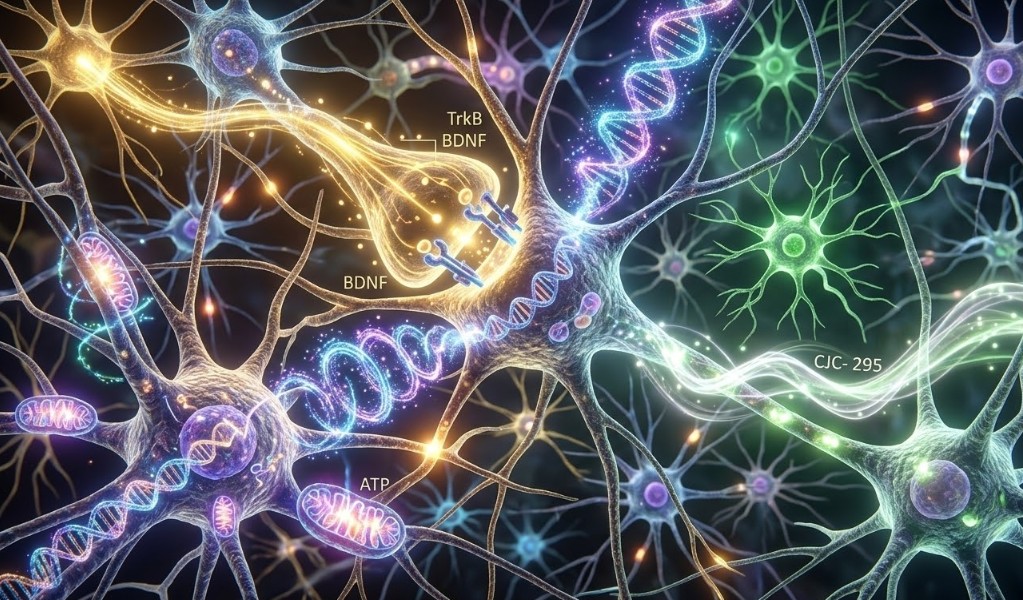
The mechanistic diversity among cognitive research peptides creates a rationale for combination protocols targeting multiple CNS biology pathways simultaneously. The Semax + NAD+ combination, for example, addresses BDNF-mediated synaptic plasticity (Semax) alongside the neuronal metabolic and DNA repair substrate that determines whether synaptic remodeling can be sustained (NAD+). The Energizer Bunny NAD+, Semax, and CJC-1295 research compound combines these mechanisms with GH axis stimulation for a multi-system research protocol.
Semax and Selank represent a different type of combination rationale: partially overlapping but distinct mechanisms that together address both the synaptic growth signal (Semax → BDNF) and the stress-induced signal that suppresses hippocampal neurogenesis (Selank → reduced HPA activation). In research models where cognitive deficit is driven by both inadequate neurotrophic support and excessive stress signaling, combining compounds targeting each mechanism independently tests whether the combination produces additive or synergistic effects.
For a broader research overview of combining cognitive and other peptide classes for multi-system CNS research, see our Peptide Stacking: Research Guide to Synergistic Laboratory Combinations. For a specific biohacking-context discussion of NAD+, Semax, and CJC-1295 multi-compound approaches, see Biohacking Research: NAD+, Semax, and CJC-1295 Multi-Compound Approaches.
Research-Grade Quality for Cognitive and Nootropic Peptides
CNS research places particular demands on compound quality. The blood-brain barrier concentration of a research compound is always lower than its plasma concentration — typically by a factor of 10 to 100 or more, depending on the compound’s BBB penetrance. This means that impurities present at the 1–2% level in plasma may be essentially absent in the CNS, but it also means that the target compound’s CNS concentration is a fraction of the administered dose. At these concentrations, trace contaminants with competing biological activity become proportionally more consequential.
For nootropic peptide research specifically:
- HPLC purity ≥98%: The minimum standard. For compounds like Dihexa, which demonstrates activity at very low concentrations in published models, the impurity contribution at sub-98% purity could represent a meaningful fraction of the total biological signal at research concentrations.
- Mass spectrometry confirmation: Particularly important for peptidomimetics like Dihexa, whose modified backbone (N-hexanoic-Tyr-Ile) and non-standard amide capping require verification that synthesis and deprotection were completed correctly. Standard HPLC cannot detect all structural isomers that might arise from racemization of the Ile residue.
- CoA documentation: Batch-specific certificate with synthesis date, purity, and mass data. For multi-compound research protocols, CoA documentation for each compound enables traceback if results are anomalous.
- Intranasal formulation purity: Semax and related compounds are often administered intranasally in research models. The nasal mucosa is a relatively permeable barrier, and excipients or impurities in the formulation vehicle can be absorbed directly. Research protocols using intranasal routes should verify excipient composition and pH of the reconstituted solution.
Spartan Peptides supplies Semax, Pinealon, and NAD+ through the Focus & Clarity research catalog at ≥98% HPLC purity with full CoA documentation. For guidance on cognitive peptide research protocols and combination design, see Cognitive Peptide Guide: Semax, Selank, and Dihexa Research.
Frequently Asked Questions
Q: How does Semax produce BDNF elevation, and why does this matter for cognitive research?
Semax is thought to increase BDNF expression through a combination of ACTH receptor (MC4R) partial agonism — which elevates cAMP and activates PKA, a transcription factor regulator that drives BDNF gene expression — and possible direct effects on serotonergic neurons that secondarily increase BDNF production. In the hippocampus, elevated BDNF binds TrkB receptors on synapses, activating CREB phosphorylation and downstream synthesis of proteins required for long-term potentiation and synaptic strengthening. BDNF is the most widely studied mediator of activity-dependent synaptic plasticity, so compounds that raise hippocampal BDNF are studied as tools for understanding the molecular basis of learning and memory. The relevance for cognitive research is direct: BDNF deficiency is associated with reduced neuroplasticity, and its restoration is a well-characterized mechanism for improving hippocampal function in preclinical models.
Q: What is the difference between Selank’s anxiolytic mechanism and that of benzodiazepines?
Benzodiazepines act by binding an allosteric site on GABA-A receptors and increasing the frequency of chloride channel opening in response to GABA, a direct positive modulation of the receptor. Chronic benzodiazepine exposure leads to GABA-A receptor downregulation and tolerance. Selank, by contrast, acts primarily by inhibiting enkephalinase — the enzyme that degrades endogenous enkephalin peptides — thereby prolonging the activity of endogenous anxiolytic signals without direct receptor binding. It also has secondary effects on GABA-A receptor subunit expression, but does not appear to cause the receptor downregulation associated with chronic benzodiazepine use in published models. The research distinction is relevant for studies where tolerance, withdrawal, or receptor regulation are variables of interest, since Selank’s indirect mechanism produces a different receptor adaptation profile than direct allosteric modulators.
Q: Why does Dihexa’s potency comparison to BDNF require careful interpretation?
The seven-orders-of-magnitude potency comparison between Dihexa and BDNF from Benoist et al. (2013) is frequently cited but requires context. Dihexa and BDNF act on different receptors — Dihexa potentiates HGF/Met signaling, while BDNF acts on TrkB — so the comparison is between two different synaptic pathways, not between competing agonists at the same site. Additionally, the comparison was made across different administration routes and assay conditions, which introduces variables that would not be present in a direct dose-response comparison within the same in vitro system. The more appropriate interpretation is that Dihexa is active at low concentrations in the tested paradigms and that its HGF/Met mechanism produces measurable cognitive effects through a pathway independent of direct neurotrophin receptor activation. The potency figure is informative about its pharmacological efficiency, not a direct competition claim against BDNF.
Q: How does NAD+ depletion specifically affect neuronal function beyond general metabolic effects?
Neurons have specific vulnerabilities to NAD+ depletion that peripheral tissues do not share. First, neurons are post-mitotic — they cannot divide to dilute accumulated DNA damage, so PARP1-dependent DNA repair is their primary mechanism for managing genotoxic stress. When NAD+ is depleted, PARP1 activity falls, unrepaired strand breaks accumulate, and the DNA damage response can trigger apoptosis or pathological NAD+ depletion cascades (parthanatos). Second, SIRT1 in neurons drives BDNF expression and neuroprotective gene programs, so NAD+ depletion impairs not only mitochondrial function but also the neurotrophic signaling that maintains synaptic density. Third, NAD+ is required for axonal transport energy (via ATP from oxidative phosphorylation), and axonal transport failure is an early feature of several neurodegenerative conditions. The combination of these neuron-specific vulnerabilities makes NAD+ repletion research particularly relevant in the context of aging and neurodegeneration models.
Q: Is there human data for any of these cognitive research peptides?
The human data landscape varies significantly across these compounds. Semax has the most human-context data: it is registered as a medical product in Russia, and Russian literature includes controlled studies in stroke and cognitive aging, though these have not been replicated in Western trials meeting contemporary standards. Selank has one published small human trial (Semenova et al., 2010) in generalized anxiety disorder. NAD+ has the most rigorous Western human data, including randomized controlled trials documenting NAD+ elevation following precursor supplementation and a trial showing skeletal muscle insulin sensitization (Yoshino et al., 2021), though CNS-specific human outcome data remain sparse. Pinealon and Dihexa have no published human clinical trial data. The absence of controlled human data does not diminish the research value of these compounds for laboratory investigation, but it is the correct characterization of where the evidence currently stands.
Conclusion
The cognitive peptide research field is defined by mechanistic precision across several independent CNS biology pathways. Semax and Selank address neurotrophic signaling and the anxiety-cognition interface, respectively, with the most developed Russian preclinical and limited human literature of any compounds in this group. Pinealon targets neuronal gene regulation and neuroprotection through an epigenetic mechanism that is still being characterized. Dihexa’s HGF/Met mechanism represents a distinct synaptic biology pathway with striking reported potency in rodent cognitive paradigms and a clear need for independent replication. NAD+ addresses the metabolic substrate that sustains all of the above — neuronal bioenergetics, DNA repair, and SIRT1-mediated gene regulation — through mechanisms that are among the most thoroughly characterized in the aging neuroscience literature.
Spartan Peptides supplies Semax, Pinealon, and NAD+ through the Focus & Clarity research catalog at ≥98% HPLC-verified purity with full CoA documentation. For a broad overview of neuroprotection research across peptide classes, see Peptides for Neuroprotection: From Brain Fog to Brain Health.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
- Dmitrieva, V.G. et al. (2010). Effect of Semax and Selank on the expression of BDNF and its receptor TrkB after focal ischemia of the cerebral cortex in rats. Journal of Molecular Neuroscience, 41(2), 253–261. (Not indexed in PubMed)
- Semenova, T.P. et al. (2010). Selank and short peptide analogues of tuftsin modify the behavior of rats in Porsolt’s test. Bulletin of Experimental Biology and Medicine, 150(4), 415–417. (Not indexed in PubMed)
- Benoist, C.C. et al. (2013). Facilitation of hippocampal synaptogenesis and spatial memory by C-terminal truncated Nle1-angiotensin IV analogues. Journal of Pharmacology and Experimental Therapeutics, 347(2), 374–380. PMID: 23055539
- Khavinson, V.Kh. et al. (2012). Regulatory peptide Pinealon increases cell viability under conditions of oxidative stress. Advances in Gerontology, 25(4), 605–611. (Not indexed in PubMed)
- Yoshino, M. et al. (2021). Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science, 372(6547), 1224–1229. PMID: 33888596
- Belenichev, I.F. et al. (2021). Neuroprotection and neuroplasticity. Frontiers in Neuroscience, 15, 711703. (Not indexed in PubMed)
- Hashimoto, K. & Panchenko, V.A. (2011). Mechanisms of synaptic transmission modulation by brain-derived neurotrophic factor. Current Neuropharmacology, 9(4), 598–603. (Not indexed in PubMed)
- Meijer, A.J. & Codogno, P. (2007). AMP-activated protein kinase and autophagy. Autophagy, 3(3), 238–240. PMID: 17224623
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →