Complete Guide to Weight Loss Peptides
Written bySpartan Research Team

Interest in peptides for weight loss has accelerated significantly over the past five years, driven in large part by the clinical performance of GLP-1 receptor agonists and a broader expansion of metabolic peptide research. Whereas earlier weight-management pharmacology was dominated by appetite suppressants and thermogenic agents with poor tolerability profiles, peptide-based compounds now represent one of the most actively studied frontiers in obesity and metabolic disease science.
This guide provides a comprehensive, research-grounded overview of the best peptides for weight loss currently under investigation. It covers the primary mechanisms through which these compounds influence energy balance, reviews the key clinical and preclinical evidence for each, and explains what differentiates the major compound classes. Whether the research focus is GLP-1 receptor agonism, growth hormone secretagogue activity, or mitochondrial regulation, each compound class operates through a distinct and well-characterized biochemical pathway.
All compounds discussed here are research peptides. They are not approved for human consumption and are referenced strictly in the context of ongoing scientific investigation.
What Are Weight Loss Peptides?
Peptides are short chains of amino acids — typically 2 to 50 residues in length — that function as signaling molecules within biological systems. Unlike small-molecule drugs, which often act through broad receptor blockade or enzyme inhibition, peptides typically operate through highly specific receptor interactions, which has made them attractive targets in metabolic research.
The category of peptides for weight loss is not a single pharmacological class. It encompasses several mechanistically distinct compound families that converge on one or more of the following physiological processes:
- Energy intake regulation: suppressing appetite or prolonging satiety through hypothalamic or gut-brain axis signaling
- Lipid metabolism: directly targeting adipocyte lipolysis or inhibiting lipogenesis
- Growth hormone axis modulation: stimulating GH secretion, which drives downstream lipolytic activity via IGF-1 and free fatty acid release
- Mitochondrial and metabolic efficiency: improving cellular energy utilization and insulin sensitivity
Research in this area has been substantially shaped by the clinical development of GLP-1 receptor agonists — a class that includes semaglutide and, in the dual-agonist category, tirzepatide — but the preclinical literature extends well beyond these compounds to include fragment peptides, growth hormone-releasing hormones, and mitochondrial-targeted molecules.
How Weight Loss Peptides Work: Primary Mechanisms
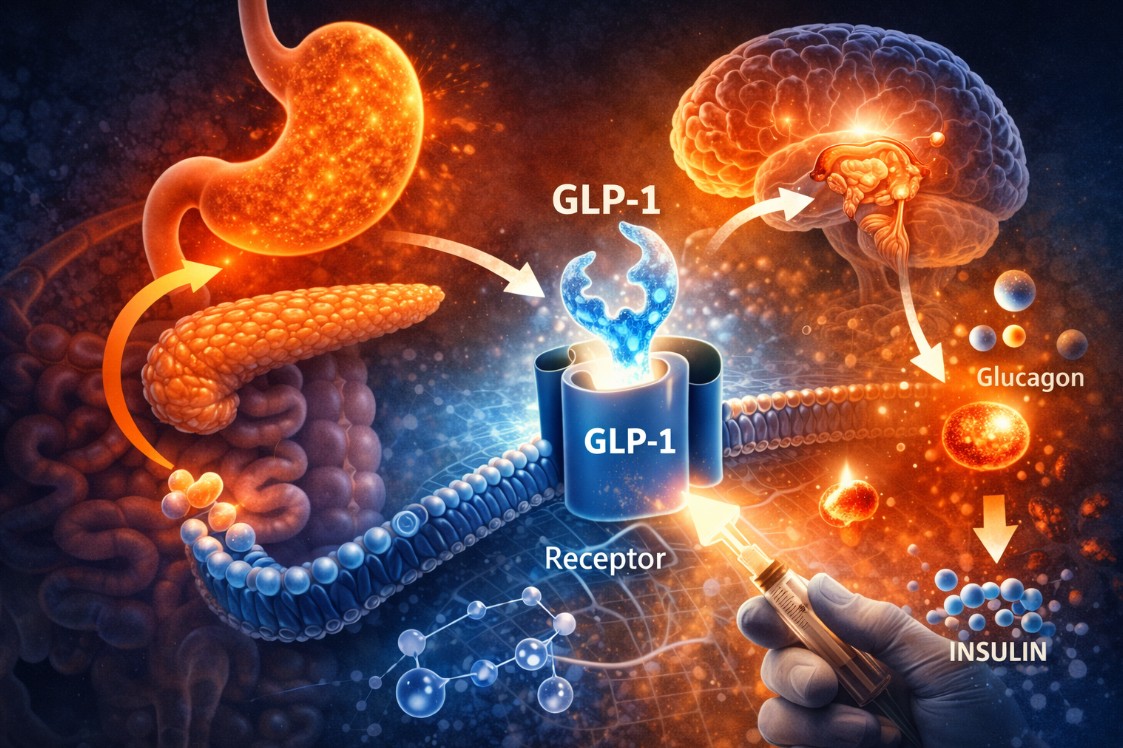
GLP-1 Receptor Agonism
Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by intestinal L-cells in response to nutrient ingestion. It acts on GLP-1 receptors in the pancreas, hypothalamus, brainstem, and gastrointestinal tract to produce several metabolically relevant effects: enhanced glucose-dependent insulin secretion, suppressed glucagon release, delayed gastric emptying, and reduced appetite signaling through hypothalamic satiety centers.
GLP-1 receptor agonist peptides are designed to mimic and extend the action of endogenous GLP-1, which has a native half-life of under two minutes due to rapid degradation by dipeptidyl peptidase-4 (DPP-4). Research-grade analogues are engineered for extended receptor occupancy, enabling the sustained metabolic effects observed in clinical trials.
Growth Hormone Secretagogue Activity
A separate and well-researched mechanism involves peptides that stimulate endogenous growth hormone (GH) secretion. GH exerts direct lipolytic effects by stimulating hormone-sensitive lipase in adipocytes, increasing free fatty acid availability for oxidation. The growth hormone-releasing hormone (GHRH) analogues and GHRP compounds achieve this through distinct receptor pathways — GHRH receptors in the anterior pituitary and ghrelin receptors, respectively — but both converge on GH pulse amplification.
Research in this category has examined compounds including tesamorelin (a GHRH analogue), CJC-1295 (a modified GHRH with extended half-life), and ipamorelin (a selective GHRP). The lipolytic effects are most consistently documented in visceral adipose tissue, which expresses relatively high GH receptor density.
Direct Lipolysis Peptides
AOD-9604 represents a distinct mechanistic class: a fragment of human growth hormone (hGH176-191) that retains the lipolytic activity of the C-terminal region while lacking the anabolic and diabetogenic properties of full-length hGH. Research indicates the compound activates fat cell beta-3 adrenergic receptors and inhibits lipogenesis, effects that appear to be independent of IGF-1 signaling.
Mitochondrial and Metabolic Regulation
MOTS-c is a peptide encoded within the mitochondrial genome — specifically from the 12S rRNA gene — that regulates cellular metabolic homeostasis. Research has documented its role in activating AMP-activated protein kinase (AMPK) signaling, improving skeletal muscle glucose uptake, and enhancing metabolic flexibility. In the context of obesity research, MOTS-c administration in murine models has been associated with resistance to diet-induced obesity and improved insulin sensitivity (Lee et al., 2015).
Key Peptides for Weight Loss: Compound Profiles
- GLP-1 Semaglutide
Semaglutide is a GLP-1 receptor agonist with approximately 94% structural homology to native GLP-1, modified with an albumin-binding fatty acid side chain that extends its plasma half-life to approximately seven days. At the receptor level, it produces the full range of GLP-1 agonist effects described above, with central appetite suppression — mediated through hypothalamic and area postrema GLP-1 receptors — representing the primary mechanism behind observed body weight reductions in research subjects. For detailed research data on this compound, see our GLP-1 Sema research guide.
Weekly semaglutide 2.4 mg subcutaneous administration in adults with obesity (without diabetes) produced a mean weight reduction of 14.9% over 68 weeks, compared with 2.4% in the placebo group. Mechanistically, imaging studies have confirmed that reductions in food intake — rather than increases in energy expenditure — account for the majority of the observed weight loss.
- GLP-2 Tirzepatide
Tirzepatide is a dual GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 receptor agonist that represents the first approved member of the ‘twincretin’ class. GIP receptor co-agonism appears to potentiate the weight loss effects of GLP-1 receptor activation through additive hypothalamic signaling, enhanced lipid clearance, and possible central nervous system effects distinct from those of GLP-1 alone. For a head-to-head comparison of these mechanisms, see our GLP-1 vs GLP-2 comparative research article.
The SURMOUNT program produced the largest pharmacologically induced weight loss data in the clinical literature at the time of publication. In SURMOUNT-1 (Jastreboff et al., 2022), participants receiving the highest tirzepatide dose achieved a mean weight reduction of 22.5% — substantially exceeding outcomes documented with GLP-1 mono-agonism. This has generated significant research interest in the additive or synergistic value of multi-receptor engagement strategies.
- Retatrutide (GLP-3)
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors — the first compound in what researchers informally classify as the GLP-3 or ‘triagonist’ class. Glucagon receptor co-agonism adds a thermogenic component absent from the dual-agonist class, as glucagon directly stimulates brown adipose tissue activity and hepatic lipid oxidation. Phase 2 trial data published in 2023 (Jastreboff et al., 2023) reported mean weight reductions of up to 24.2% over 48 weeks at the highest dose tested — the largest short-term weight loss documented in any pharmacological trial to that date. For the mechanistic research, see how GLP-3 Retatrutide works.
Retatrutide’s glucagon receptor component introduces additional complexity in research design, as glucagon agonism independently affects hepatic glucose output and must be balanced against GLP-1-mediated insulin secretion to avoid net glycemic disturbance. This three-way receptor balancing act is a central focus of ongoing mechanistic research.
For a direct comparison between tirzepatide and retatrutide research data, see GLP-3 Reta vs GLP-2 Tirz.
- AOD-9604
AOD-9604 (Advanced Obesity Drug 9604) is the C-terminal fragment of human growth hormone, comprising residues 176–191. It was originally developed by Metabolic Pharmaceuticals as an anti-obesity agent, having been isolated from hGH specifically because early research suggested the lipolytic properties of hGH resided in this region. Unlike full-length hGH, AOD-9604 does not appear to stimulate IGF-1 production or affect insulin sensitivity in preclinical models, which substantially simplifies its research safety profile.
In vitro and animal studies have demonstrated that AOD-9604 stimulates lipolysis in fat cells and inhibits lipogenesis — the synthesis of new fat — through mechanisms involving beta-3 adrenergic receptor activation and possible TGF-beta pathway modulation. Phase 2 human clinical trials conducted during the compound’s pharmaceutical development phase showed modest but consistent reductions in body weight relative to placebo, though the compound did not advance to Phase 3, and current interest is primarily in its research utility as a clean lipolytic model compound.
Spartan Peptides supplies AOD-9604 research peptide at ≥98% purity with HPLC verification.
- Tesamorelin
Tesamorelin is a synthetic analogue of growth hormone-releasing hormone (GHRH) stabilized by the addition of a trans-3-hexenoic acid group at the N-terminus, which protects against endopeptidase cleavage and extends functional half-life. It stimulates pulsatile GH secretion from the anterior pituitary by binding GHRH receptors, which in turn drives hepatic IGF-1 production and downstream lipolytic effects in adipose tissue.
Tesamorelin holds the distinction of being the only GHRH analogue to have received FDA approval for a specific indication: reduction of excess abdominal fat (lipodystrophy) in HIV-infected patients receiving antiretroviral therapy. In that clinical context, Phase 3 trials documented significant reductions in visceral adipose tissue area by MRI assessment over 26 weeks (Falutz et al., 2010). The visceral fat specificity is notable and reflects the relatively high GH receptor expression in visceral versus subcutaneous adipose depots.
In non-clinical obesity research models, tesamorelin’s effects are consistently focused on visceral fat rather than subcutaneous compartments, which is mechanistically consistent with its GH-mediated action profile.
Explore Tesamorelin 5mg research compound and the Spartan Strong CJC-Tesamorelin blend for research combination protocols.
- CJC-1295 / Ipamorelin
CJC-1295 is a GHRH analogue modified with drug affinity complex (DAC) technology, allowing covalent binding to albumin and extending its half-life to approximately 6–8 days — substantially longer than native GHRH’s 7-minute half-life. This enables sustained stimulation of GH pulses rather than the acute single-pulse response of shorter GHRH analogues. Ipamorelin is a selective growth hormone secretagogue receptor (GHSR) agonist derived from the GHRP class, notable for its selectivity — it stimulates GH release with minimal co-stimulation of ACTH, cortisol, or prolactin, which has made it a preferred GHRP in research settings.
When combined, CJC-1295 and ipamorelin act on complementary receptor pathways — GHRH receptors and ghrelin/GHSR receptors, respectively — to produce synergistic GH pulse amplification. Animal studies using the combination have demonstrated significant increases in GH and IGF-1 levels, with associated reductions in adipose tissue and increases in lean body mass in some models.
Spartan Peptides offers the CJC-1295 / Ipamorelin 10mg blend for research use.
- MOTS-c
MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a mitochondria-derived peptide that has attracted significant research interest for its role in systemic metabolic regulation. Unlike the peptides discussed above — which all operate through extracellular receptor signaling — MOTS-c translocates to the nucleus under conditions of metabolic stress, where it regulates gene expression related to glucose metabolism, fatty acid utilization, and antioxidant defense. Full mechanistic and mitochondrial research is available in our MOTS-c and Mitochondrial Health article.
The foundational MOTS-c research (Lee et al., 2015, Cell Metabolism) demonstrated that exogenous MOTS-c administration in mice fed a high-fat diet significantly attenuated weight gain, improved insulin sensitivity, and enhanced skeletal muscle glucose uptake via AMPK activation. At the pathway level, MOTS-c appears to regulate the folate cycle and de novo purine biosynthesis, affecting AICAR levels upstream of AMPK — a mechanistic pathway distinct from all other weight-relevant peptides discussed in this guide.
Comparative Overview: Weight Loss Peptides at a Glance
| Compound | Primary Mechanism | Key Research Finding | Receptor Target | Compound Class |
|---|---|---|---|---|
| GLP-1 Sema | GLP-1 receptor agonism | ~15% body weight reduction (STEP 1, 2021) | GLP-1R | Incretin analogue |
| GLP-2 Tirz | Dual GIP + GLP-1 agonism | ~22.5% weight reduction (SURMOUNT-1, 2022) | GLP-1R / GIPR | Twincretin |
| Retatrutide | Triple GLP-1/GIP/Glucagon | ~24.2% weight reduction (Phase 2, 2023) | GLP-1R / GIPR / GcgR | Triagonist |
| AOD-9604 | Direct lipolysis / lipogenesis inhibition | Fat-selective; no IGF-1 effect in models | Beta-3 AR | HGH fragment |
| Tesamorelin | GHRH receptor agonism → GH release | Visceral fat reduction — FDA-approved indication | GHRHR | GHRH analogue |
| CJC-1295 / Ipa | GHRH + GHSR agonism → GH amplification | Synergistic GH pulse increase in animal models | GHRHR + GHSR | GHRH + GHRP blend |
| MOTS-c | AMPK activation, mitochondrial regulation | DIO resistance; insulin sensitivity improvement (2015) | Nuclear / AMPK pathway | Mitokine |
Research Evidence: What the Clinical and Preclinical Data Show
GLP-1 and Dual Agonist Trials
The most extensive clinical evidence base in weight-loss peptide research belongs to the GLP-1 agonist class, driven by pharmaceutical development programs for type 2 diabetes and obesity indications. The STEP trials (semaglutide, 2021) and SURMOUNT trials (tirzepatide, 2022–2023) represent landmark datasets documenting percentage weight loss at a scale not previously achieved with any non-surgical intervention. These trials used rigorously controlled Phase 3 designs with 68–72 week follow-up periods, providing high-quality evidence for the magnitude and durability of effect.
Key observations from this literature include: (1) weight loss is predominantly driven by reduced caloric intake rather than increased energy expenditure; (2) lean mass is also reduced alongside fat mass, though the ratio favors fat loss; (3) effects reverse substantially upon compound discontinuation, indicating ongoing administration is required to maintain metabolic effects; and (4) gastrointestinal adverse effects are the primary tolerability concern in clinical populations.
Growth Hormone Secretagogue Research
Tesamorelin’s clinical evidence base is defined by the Phase 3 Egrifta trials in HIV-associated lipodystrophy, which consistently demonstrated 15–20% reductions in visceral adipose tissue area by MRI over 26 weeks. The compound’s lipodystrophy approval provides a model for understanding how GHRH receptor agonism selectively affects visceral fat — a finding with potential relevance to broader metabolic research even outside the HIV context.
CJC-1295 and ipamorelin data are predominantly derived from animal studies and small human pharmacokinetic trials. The human data document robust GH and IGF-1 elevation following administration, but controlled trials examining body composition as a primary endpoint in obesity models are limited. Most body composition claims in the research literature are extrapolated from GH physiology rather than directly demonstrated in controlled human studies.
AOD-9604 Clinical Data
AOD-9604 underwent formal clinical development, with Phase 2 trials conducted in overweight and obese subjects. Meta-analysis of these trials (Heffernan et al., 2001; subsequent Phase 2 data) showed modest but statistically significant weight loss relative to placebo at mid-range doses. The compound was not advanced to Phase 3 development, and its current status is as a research tool rather than a clinical development candidate. The clean separation between its lipolytic activity and IGF-1/insulin-related effects makes it a useful model compound for investigating direct adipocyte biology.
MOTS-c Preclinical Data
MOTS-c research remains primarily preclinical, anchored by the 2015 Cell Metabolism publication and subsequent work from the same group examining exercise mimicry and age-related metabolic decline. In murine high-fat diet models, MOTS-c administration consistently attenuates weight gain, reduces adipose tissue accumulation, and improves glucose tolerance. Mechanistic studies have traced these effects through the AICAR-AMPK pathway, which intersects with multiple downstream targets, including fatty acid oxidation, mitochondrial biogenesis, and glucose transporter expression. Human data on MOTS-c are limited but emerging, with early pharmacokinetic and tolerability work underway.
Combination Research Approaches

An increasingly active area of research involves investigating combinations of weight-loss peptides from mechanistically distinct classes. The rationale is that compounds acting through complementary pathways — for example, GLP-1 receptor agonism combined with growth hormone secretagogue activity — may produce additive or synergistic metabolic effects while potentially allowing lower individual doses. Spartan Peptides’ Skinny Fit weight management stack covers current research thinking on multi-compound approaches.
The triagonist retatrutide itself exemplifies this logic applied at the molecular level — simultaneous engagement of GLP-1, GIP, and glucagon receptors within a single compound architecture. The additive weight loss observed in Phase 2 trials (24.2% mean) relative to the dual-agonist class (~22.5%) supports the principle that extending receptor coverage can incrementally improve metabolic outcomes, though the relationship is likely not linear.
At the preclinical level, combinations involving GH secretagogues and direct lipolytic peptides have been examined in animal models, with some evidence of complementary effects on lean mass preservation and fat oxidation. Research in this area is active but early-stage relative to the GLP-1 agonist literature. For a broader view, see multi-compound metabolic research approaches.
Research-Grade Compound Quality: What the Literature Requires
Research reproducibility in peptide science depends critically on compound quality. Purity, correct sequence verification, and absence of interfering contaminants are prerequisites for interpretable data, and the peptide supply chain has historically been a source of significant variability across research groups.
The key quality parameters for weight-loss peptide research compounds are:
- Purity ≥98% by HPLC: Sub-98% purity introduces the possibility that observed effects — or lack thereof — are attributable to impurities rather than the compound of interest. High-performance liquid chromatography with UV detection at 214 or 220nm is the standard analytical method.
- Mass spectrometry verification: Confirms molecular weight matches the theoretical value for the target sequence, ruling out truncation, oxidation, or incorrect synthesis.
- Certificate of Analysis (CoA): A batch-specific document reporting actual purity, mass spectrometry data, and synthesis date. Research protocols typically require CoA documentation for traceability.
- Endotoxin testing: For in vivo research applications, bacterial endotoxin contamination is a confounding variable that must be controlled. Limulus amebocyte lysate (LAL) testing is standard.
Frequently Asked Questions
Q: What is the difference between GLP-1 agonists and growth hormone secretagogues in weight loss research?
GLP-1 receptor agonists reduce body weight primarily by suppressing appetite through central and peripheral satiety signaling, leading to significant reductions in caloric intake. Their effects are well-documented in large clinical trials. Growth hormone secretagogues, by contrast, work downstream through GH-driven lipolysis, particularly in visceral adipose tissue, and have a stronger body composition dimension — tending to reduce fat while preserving or increasing lean mass. These are mechanistically distinct approaches that target different physiological processes; in research models, they can be studied independently or in combination to examine interaction effects.
Q: Why does retatrutide show greater weight loss than semaglutide in clinical data?
Retatrutide engages three receptors — GLP-1, GIP, and glucagon — compared to semaglutide’s single GLP-1 receptor target. Each additional receptor engagement adds a mechanistically independent contribution: GIP receptor co-agonism potentiates hypothalamic satiety signaling and improves lipid handling, while glucagon receptor co-agonism adds a thermogenic component through brown adipose tissue activation and hepatic lipid oxidation. The cumulative effect of these three convergent mechanisms on energy balance appears to exceed what single-receptor agonism achieves. The 2023 Phase 2 trial data (Jastreboff et al., 2023) showed mean reductions of approximately 24.2% — suggesting that adding glucagon receptor engagement to a GLP-1/GIP base produces a meaningful incremental effect.
Q: What distinguishes AOD-9604 from full-length growth hormone in weight loss research?
AOD-9604 is the C-terminal fragment (residues 176–191) of human growth hormone, isolated specifically because early research attributed hGH’s lipolytic properties to this region. Full-length hGH stimulates both fat breakdown and protein anabolism through IGF-1-mediated pathways, and also carries risks of insulin resistance and glucose dysregulation with chronic administration. AOD-9604 appears to retain the lipolytic activity without generating significant IGF-1 elevation or insulin-related effects in preclinical models, making it a cleaner research tool for studying direct adipocyte biology. This mechanistic selectivity is the primary reason it attracted pharmaceutical development interest as a weight-loss compound independent of full hGH activity.
Q: How is MOTS-c mechanistically different from other peptides for weight loss?
MOTS-c is a mitochondria-derived peptide — encoded within mitochondrial DNA rather than nuclear DNA — that functions as an intracellular and systemic metabolic regulator. Unlike receptor-targeted peptides such as GLP-1 agonists or GHRH analogues, MOTS-c translocates to the nucleus under metabolic stress and directly modulates gene transcription related to energy metabolism. Its downstream effects — AMPK activation, improved glucose uptake, enhanced fatty acid oxidation — emerge from this transcriptional regulation rather than cell-surface receptor binding. This makes MOTS-c uniquely positioned as a mitochondrial health research tool rather than a classical weight-loss receptor agonist.
Q: What should researchers look for when sourcing weight loss research peptides?
The minimum quality requirements for interpretable research are: HPLC-verified purity ≥98%, mass spectrometry confirmation of correct molecular weight, batch-specific Certificate of Analysis documentation, and US-origin shipping for intact cold-chain management. For in vivo research, endotoxin testing data add an important additional layer of quality control. Sourcing from suppliers with published quality standards and transparent documentation — rather than opaque bulk suppliers — is essential for data reproducibility and protocol integrity.
Conclusion
Research into peptides for weight loss has produced one of the most rapidly evolving bodies of evidence in contemporary metabolic science. From the GLP-1 agonist class — now with Phase 3 clinical data documenting weight reductions exceeding 20% of body weight — to growth hormone secretagogues, direct lipolytic fragments, and mitochondrial regulators, the mechanistic landscape is remarkably diverse. Each compound class illuminates a different facet of energy homeostasis biology, and the most productive research programs increasingly examine combinations and interactions across these classes.
For researchers working in this space, Spartan Peptides maintains a dedicated weight loss research peptide catalog covering GLP-1 Sema, GLP-2 Tirzepatide, Retatrutide, AOD-9604, Tesamorelin, CJC-1295/Ipamorelin, and MOTS-c — all supplied at ≥98% HPLC-verified purity with full Certificate of Analysis documentation.
For next-generation compound research, see our overview of the future of weight loss peptides.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
Heffernan, M.A. et al. (2001). An analog of the human growth hormone fragment AOD9604 retains the anti-obesity activity of the parent hormone but has a reduced diabetogenic activity. JPET, 297(1), 241–246.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →