Kisspeptin vs Enclomiphene: Mechanisms, Research, and Key Differences
Written bySpartan Research Team
Kisspeptin and enclomiphene both come up regularly in reproductive endocrinology research. The overlap stops there. These are fundamentally different compounds targeting different points in the HPG axis with different pharmacological profiles entirely. One is an endogenous neuropeptide that triggers GnRH neuron firing from the very top of the hypothalamic signaling cascade. The other is a synthetic small molecule that blocks estrogen receptor feedback two steps downstream.
That distinction matters for study design. If you’re investigating upstream signaling through KISS1R or GnRH pulse dynamics, kisspeptin is the more direct research tool. If you’re looking at estrogen feedback modulation or testosterone restoration in hypogonadal models, enclomiphene has the deeper clinical dataset. We reviewed the published mechanisms and research profiles for both compounds below.
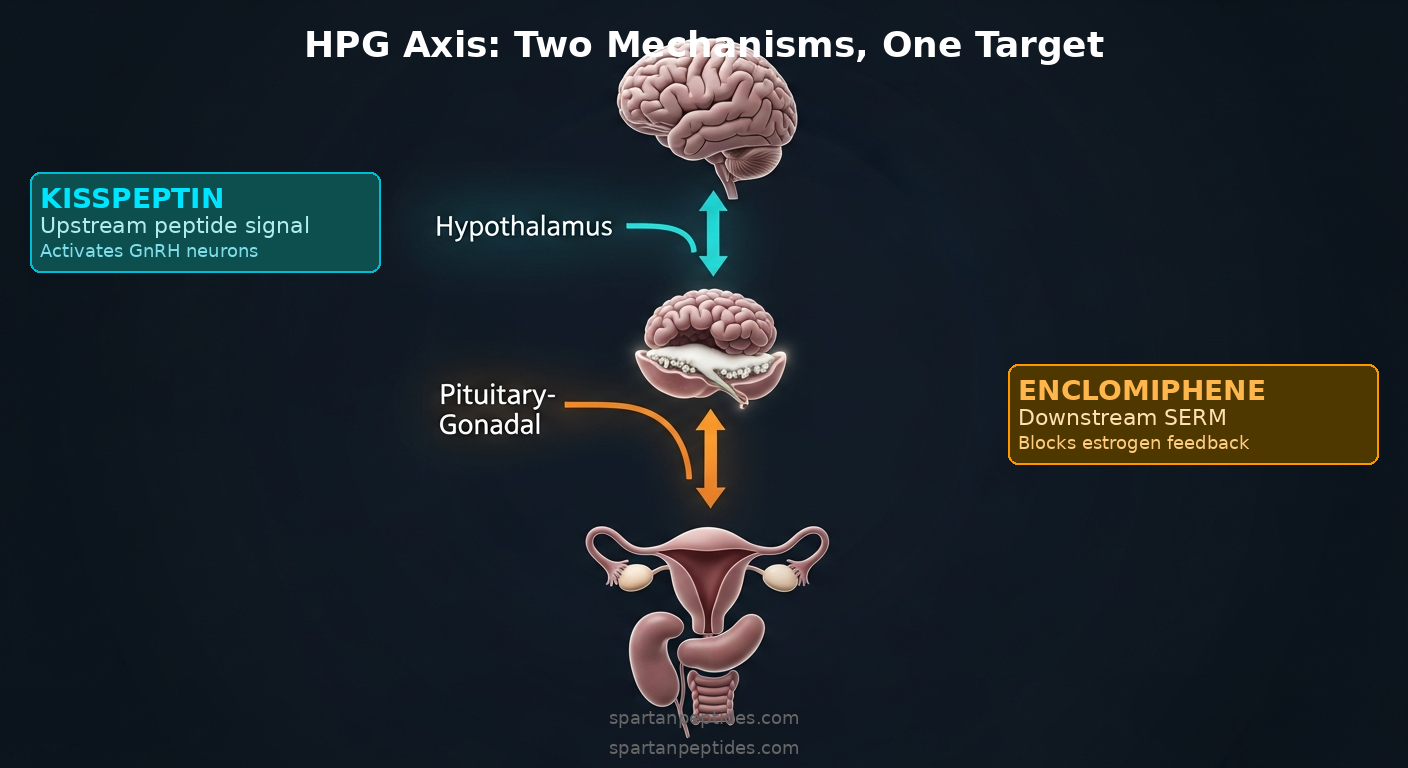
Kisspeptin: Mechanism and Research Profile
How Kisspeptin Signals Through the HPG Axis
Kisspeptin is a neuropeptide encoded by the KISS1 gene. It binds to the KISS1R receptor (also known as GPR54), expressed on GnRH neurons in the hypothalamus. And it’s remarkably potent as a GnRH secretion stimulus. That’s what makes it useful as a research tool: it acts at the origin point of the reproductive hormone cascade, before GnRH itself fires.
When kisspeptin activates KISS1R, GnRH neurons discharge and release GnRH into the hypophyseal portal system. The anterior pituitary responds by secreting LH and FSH. Gonadal stimulation and sex steroid production follow downstream. So kisspeptin doesn’t interact with estrogen receptors or gonadotropins directly. It works one step earlier than that.
Key Research Findings
The discovery of kisspeptin’s role in reproductive signaling came from observations that loss of function mutations in KISS1R caused hypogonadotropic hypogonadism in both humans and animal models (de Roux et al., 2003; PMID: 14573733). This established kisspeptin signaling as essential for normal reproductive function.
Dhillo et al. (2005) demonstrated that intravenous kisspeptin administration in healthy male volunteers produced robust increases in plasma LH, FSH, and testosterone (PMID: 16234304). This was among the first human studies confirming kisspeptin’s gonadotropin releasing activity.
Jayasena et al. (2011) expanded on this work, showing that kisspeptin administration could stimulate gonadotropin release in women across different phases of the menstrual cycle, with particularly strong LH responses during the preovulatory phase (PMID: 21976724).
More recent investigations have explored kisspeptin in the context of reproductive endocrinology and fertility protocols. Abbara et al. (2015) published data showing kisspeptin could trigger oocyte maturation in IVF settings while potentially reducing the risk of ovarian hyperstimulation syndrome (PMID: 26227985).
Research Areas and Applications
Kisspeptin research spans several domains within reproductive science:
- LH and FSH pulsatility regulation and GnRH neuron physiology
- Fertility research, including IVF trigger protocols
- Hypothalamic amenorrhea and hypogonadotropic hypogonadism models
- Neuroendocrine control of puberty onset
- Sexual arousal and limbic system activation (Comninos et al., 2017; PMID: 28060925)
As an endogenous signaling peptide, kisspeptin acts through the body’s own receptor systems. It does not block or antagonize any receptor. Instead, it activates a natural signaling pathway, which is a fundamentally different pharmacological approach compared to SERMs.
Enclomiphene: Mechanism and Research Profile
How Enclomiphene Modulates Estrogen Feedback
Enclomiphene is the trans isomer of clomiphene citrate. It’s a SERM, specifically antagonizing estrogen receptors in the hypothalamus and pituitary. By blocking estrogen’s negative feedback, it effectively convinces the HPG axis it’s operating in a low-estrogen state. That perception triggers increased GnRH and gonadotropin secretion.
The contrast with kisspeptin here is worth being precise about. Kisspeptin activates a receptor. Enclomiphene blocks one. Both can drive LH and FSH up, but through opposite pharmacological mechanisms. That’s not a small difference for researchers designing mechanistic experiments.
Key Research Findings
Kaminetsky et al. (2013) published Phase II trial data demonstrating that enclomiphene restored testosterone levels to the normal range in men with secondary hypogonadism while preserving spermatogenesis. This was a notable finding because exogenous testosterone replacement typically suppresses sperm production (PMID: 23595628).
Wiehle et al. (2014) reported results from Phase III trials showing enclomiphene increased total testosterone, LH, and FSH in hypogonadal men compared to placebo, with a favorable side effect profile compared to racemic clomiphene (PMID: 24657458).
Kim et al. (2017) examined clomiphene (which contains both enclomiphene and zuclomiphene isomers) in a systematic review of male infertility studies, finding consistent improvements in hormone parameters across multiple trials (PMID: 28029592).
Research Areas and Applications
Enclomiphene research has focused primarily on:
- Secondary hypogonadism in males, particularly testosterone restoration
- Fertility preservation during hormonal optimization protocols
- Comparative studies against testosterone replacement therapy
- SERM pharmacology and estrogen receptor selectivity
Enclomiphene is a small molecule pharmaceutical, not a peptide. It is orally bioavailable and has a longer duration of action compared to most peptide compounds. Its development has progressed through formal clinical trials, giving it a relatively mature clinical dataset for its primary indications.
Kisspeptin vs Enclomiphene: Head to Head Comparison
| Parameter | Kisspeptin | Enclomiphene |
|---|---|---|
| Compound Class | Endogenous neuropeptide | Selective estrogen receptor modulator (SERM) |
| Mechanism of Action | Activates KISS1R on GnRH neurons, stimulating GnRH release | Blocks estrogen receptors, removing negative feedback on GnRH/gonadotropin secretion |
| Primary Target | Hypothalamic GnRH neurons (upstream) | Estrogen receptors at hypothalamus and pituitary |
| Pharmacological Action | Receptor agonist (activating) | Receptor antagonist (blocking) |
| Administration Route | Subcutaneous or intravenous injection | Oral |
| Research Maturity | Growing clinical dataset, strong preclinical foundation | Multiple Phase III clinical trials completed |
| Key Research Endpoints | LH/FSH pulsatility, GnRH dynamics, fertility protocols | Testosterone restoration, spermatogenesis preservation |
| Endogenous Equivalent | Yes, naturally produced in the hypothalamus | No, synthetic pharmaceutical compound |
Key Differences for Researchers
Upstream vs Downstream HPG Axis Targeting
The most fundamental difference is where each compound acts. Kisspeptin sits at the top of the HPG cascade, directly activating neurons that initiate GnRH secretion. It’s a probe for the upstream signals that start the reproductive hormone cascade. Enclomiphene acts further downstream, modulating the estrogen feedback loop that normally fine-tunes GnRH and gonadotropin output. Different questions, different tools.
Researchers studying feedback mechanisms and estrogen receptor dynamics will find enclomiphene the more direct intervention point. Those studying GnRH neuron physiology and the biology of reproductive axis initiation will find kisspeptin harder to replace.
Peptide Signaling vs Receptor Antagonism
Kisspeptin activates a receptor. Enclomiphene blocks one. That’s worth saying plainly because it shapes how you design the experiment. Agonist approaches (kisspeptin) give you a positive stimulus you can time precisely, useful for studying acute GnRH pulse dynamics and receptor-level activation. Antagonist approaches (enclomiphene) remove an inhibitory input, producing a more sustained hormonal shift that persists past the acute dosing window.
For GnRH neuron physiology research, kisspeptin offers a direct probe. For estrogen feedback regulation and receptor selectivity studies, enclomiphene is the cleaner intervention. Related mechanistic comparisons are covered in our PT 141 vs PDE5 inhibitors research comparison. Researchers investigating melanocortin receptor-pathway peptides can also review the PT-141 compound profile or source PT-141 for research.
Combinability in Research Protocols
Because kisspeptin and enclomiphene target non overlapping mechanisms, some investigators have considered whether they can be studied in combination. Theoretically, activating GnRH neurons with kisspeptin while simultaneously removing estrogen negative feedback with enclomiphene could produce additive or synergistic effects on gonadotropin output. However, published data on this specific combination remains limited, and the interaction dynamics are not yet well characterized.
Different Research Use Cases
Kisspeptin research gravitates toward acute neuroendocrine signaling, fertility applications, and the fundamental biology of reproductive axis activation. Its endogenous nature is a genuine advantage here. You’re studying physiological regulation without introducing a pharmacologically foreign compound into the system, which matters for certain mechanistic questions.
Enclomiphene research has concentrated on testosterone optimization and fertility preservation, particularly in males with secondary hypogonadism. It’s orally bioavailable with a more established clinical trial history than most research peptides. That makes it better suited for longer-duration protocol designs where sustained hormonal shifts are the endpoint. Additional context on reproductive signaling research is covered in our PT 141 research in female models article and the broader sexual health peptides section.
Frequently Asked Questions
What is the difference between kisspeptin and enclomiphene?
Kisspeptin is an endogenous neuropeptide that activates GnRH neurons at the hypothalamic level, serving as an upstream regulator of the HPG axis. Enclomiphene is a selective estrogen receptor modulator (SERM) that blocks estrogen negative feedback at the pituitary and hypothalamus. They represent fundamentally different compound classes with distinct mechanisms for influencing gonadotropin release.
Can kisspeptin and enclomiphene be studied together?
Some research protocols have explored combination approaches targeting multiple nodes of the HPG axis simultaneously. Because kisspeptin acts upstream on GnRH neurons and enclomiphene modulates estrogen receptor feedback, their mechanisms do not directly overlap. However, combinability remains an active area of investigation with limited published data.
Is kisspeptin a SERM?
No. Kisspeptin is not a SERM. It is an endogenous neuropeptide that binds to the KISS1R (GPR54) receptor on GnRH neurons. SERMs like enclomiphene work by blocking estrogen receptors. These are entirely different pharmacological mechanisms.
Which compound has more clinical research data?
Enclomiphene has a larger body of clinical trial data in human subjects, particularly for secondary hypogonadism, including multiple Phase III trials. Kisspeptin has robust preclinical data and a growing number of human studies, especially in reproductive endocrinology and fertility research contexts.
Does kisspeptin increase testosterone in research models?
Kisspeptin administration has been documented to stimulate LH and FSH release through GnRH neuron activation, which in turn has been associated with downstream testosterone elevation in both preclinical and clinical research settings. Key studies include work by Dhillo et al. (2005, PMID: 16234304) and Jayasena et al. (2011, PMID: 21976724) demonstrating acute gonadotropin responses.
Source Research Grade Kisspeptin for Your Studies
Researchers investigating kisspeptin signaling pathways, GnRH neuron activation, or HPG axis dynamics can source research grade Kisspeptin 10 from Spartan Peptides. All compounds are third party tested for purity and identity. Browse our full Research Library for additional compound profiles and study references, or visit the Kisspeptin compound hub for a consolidated overview of available research. Researchers investigating the broader landscape of reproductive and sexual health peptides can find additional context in our complete guide.
This article was prepared by the Spartan Research Team for informational and research purposes only. All compounds referenced are intended for in vitro research use. This content does not constitute medical advice.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →