NAD+ Peptide: Research Guide to Nicotinamide Adenine Dinucleotide and Cellular Energy
Written bySpartan Research Team

Nicotinamide Adenine Dinucleotide — NAD+ — has emerged as one of the most intensively studied compounds in longevity and cellular biology research. Present in every living cell, NAD+ functions as a coenzyme in hundreds of enzymatic reactions, spanning mitochondrial energy production, DNA repair, and gene expression regulation. Researchers studying aging biology, metabolic function, and neuroprotection consistently place NAD+ at the intersection of multiple longevity pathways. This guide covers the biochemistry, preclinical research landscape, and sourcing considerations for scientists and researchers working with NAD+ in their protocols.
🔬 Key Research Findings
- NAD+ is a dinucleotide coenzyme — not technically a peptide, but widely researched alongside peptides in longevity and cellular optimization protocols
- NAD+ levels documented to decline with age due to increased CD38 and PARP activity
- Sirtuin (SIRT1-7) activation is NAD+-dependent — the NAD+/sirtuin axis is a primary target in longevity research
- PARP-mediated DNA strand break repair requires NAD+ as a substrate — critical for genomic stability research
- Injectable NAD+ provides direct cellular repletion; oral precursors (NMN, NR) require enzymatic conversion
Is NAD+ a Peptide? Understanding the Classification
The question “is NAD+ a peptide” is one of the most common queries researchers enter when first exploring this compound — and it deserves a precise answer.
By strict biochemical definition, NAD+ is not a peptide. Peptides are composed of amino acid chains linked by peptide bonds. NAD+ (Nicotinamide Adenine Dinucleotide) is a dinucleotide coenzyme — a molecule composed of adenosine monophosphate (AMP) linked to nicotinamide mononucleotide (NMN) via a phosphoanhydride bond. It belongs to the nucleotide family, not the peptide family.
So why does NAD+ appear in peptide catalogs, and why do researchers group it alongside peptides like BPC-157, GHK-Cu, and MOTS-c?
- Small bioactive molecule: Like research peptides, NAD+ is a small, biologically active molecule studied for its direct signaling and metabolic roles at the cellular level.
- Shared research contexts: NAD+ is used in the same longevity, cellular optimization, and anti-aging research protocols as research peptides — often studied in combination with them.
- Same researcher demographic: Biohackers, longevity researchers, and metabolic scientists who work with peptides typically also work with NAD+ in overlapping experimental designs.
- Supplier categorization: Many research-grade suppliers list NAD+ in their peptide catalog as a practical organizational choice, reflecting how researchers actually search for and procure it.
The classification distinction is real and scientifically important — but the grouping reflects practical research reality. NAD+ is not a peptide; it is frequently studied alongside peptides in the same research frameworks.
What Is NAD+?
NAD+ stands for Nicotinamide Adenine Dinucleotide. It is a dinucleotide coenzyme found in every living cell across all biological kingdoms. NAD+ exists in two primary forms that form a critical redox pair:
- NAD+ (oxidized form): The electron acceptor form — accepts electrons and becomes NADH during metabolic reactions.
- NADH (reduced form): The electron donor form — donates electrons to the mitochondrial electron transport chain to drive ATP synthesis.
The ratio of NAD+ to NADH serves as a critical metabolic signaling marker, reflecting the oxidative state of the cell and influencing numerous downstream enzymatic processes.
NAD+ biosynthesis pathways studied in research include:
- Preiss-Handler pathway: Synthesizes NAD+ from niacin (nicotinic acid) via the intermediate nicotinic acid mononucleotide (NAMN).
- Salvage pathway: Recycles nicotinamide (NAM) — a byproduct of NAD+-consuming reactions — back into NAD+. This pathway, involving NAMPT (nicotinamide phosphoribosyltransferase) as the rate-limiting enzyme, is the primary route of NAD+ regeneration in mammalian cells.
- De novo synthesis: NAD+ synthesized from tryptophan through the kynurenine pathway.
Key NAD+ precursors studied in research include NMN (nicotinamide mononucleotide), NR (nicotinamide riboside), and niacin — each with distinct pharmacokinetics and tissue uptake profiles. Multiple peer-reviewed studies document that endogenous NAD+ concentrations decline measurably with age across multiple tissue types, forming a central hypothesis in aging biology research.
NAD+ and Mitochondrial Function in Research
NAD+ is essential to mitochondrial oxidative phosphorylation — the process by which cells generate the majority of their ATP. In the mitochondrial electron transport chain, NADH donates electrons to Complex I (NADH:ubiquinone oxidoreductase, also called NADH dehydrogenase), initiating a cascade that drives proton pumping across the inner mitochondrial membrane and ultimately powers ATP synthase (Complex V).
Key research findings in this area include:
- Complex I dependency: Complex I is the largest and most elaborate enzyme in the electron transport chain. Its function is directly dependent on adequate NADH availability, which requires sufficient NAD+ for the upstream oxidative reactions in the TCA cycle.
- NAD+/NADH ratio as metabolic signal: The ratio functions as a real-time indicator of cellular energy status. A declining NAD+/NADH ratio is associated with impaired mitochondrial function in preclinical aging models.
- Mitochondrial biogenesis: Preclinical data suggest that NAD+ repletion supports mitochondrial biogenesis pathways, including PGC-1α activation, in animal models.
A key reference in this area: Rajman, Chwalek & Sinclair (2018), Cell Metabolism — “Therapeutic Potential of NAD-Boosting Molecules: The In Vivo Evidence” (PMID 28825891) — provides a comprehensive review of in vivo evidence for NAD+ repletion strategies across multiple organ systems and aging models.
NAD+ and Sirtuin Activation
Sirtuins are a family of seven NAD+-dependent deacetylase enzymes (SIRT1–SIRT7) that regulate a broad range of cellular processes, including gene expression, DNA repair, metabolic homeostasis, and stress response. Because sirtuins require NAD+ as a co-substrate — consuming one molecule of NAD+ per deacetylation reaction — cellular NAD+ availability directly gates sirtuin activity.
Research highlights in the NAD+/sirtuin axis include:
- SIRT1: The most studied sirtuin in longevity research. SIRT1 deacetylates histones and transcription factors including PGC-1α, FOXO proteins, and p53, influencing mitochondrial biogenesis, apoptosis, and metabolic gene programs.
- SIRT3: Localized primarily to the mitochondrial matrix. SIRT3 regulates mitochondrial protein acetylation, oxidative phosphorylation efficiency, and ROS management in preclinical models.
- Caloric restriction mimicry: Caloric restriction robustly activates sirtuins in preclinical models, in part by raising the NAD+/NADH ratio. NAD+ repletion has been studied as a potential means of activating this same axis without caloric restriction.
- Metabolic homeostasis: The NAD+/sirtuin axis modulates insulin sensitivity, lipid metabolism, and circadian rhythm regulation in animal models.
Reference: Schultz & Sinclair (2016), Cell Metabolism — “Why NAD+ Declines during Aging: It’s Destroyed” (PMID 30232201) — addresses the enzymatic consumption mechanisms that drive age-related NAD+ depletion, providing mechanistic context for sirtuin-focused research.
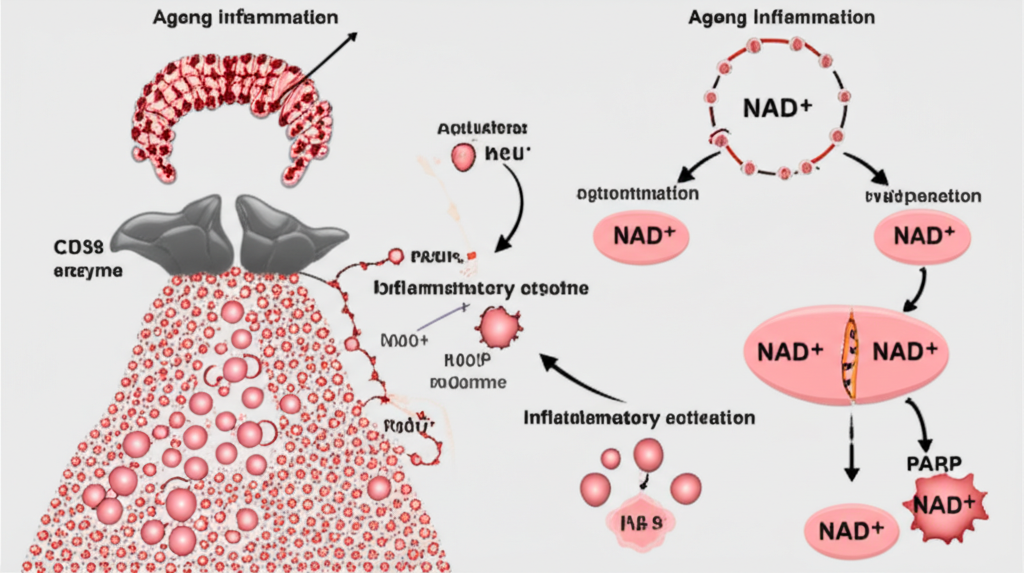
NAD+ and DNA Repair Mechanisms
NAD+ is a direct substrate for PARP enzymes (Poly ADP-ribose polymerases) — a family of nuclear proteins central to the DNA damage response. When DNA strand breaks occur, PARP enzymes are activated and consume NAD+ to synthesize poly(ADP-ribose) (PAR) chains, which serve as scaffolds for DNA repair machinery recruitment.
Key aspects studied in research models:
- PARP-mediated NAD+ consumption: In response to oxidative stress or genotoxic damage, PARP-1 activation can rapidly consume significant proportions of cellular NAD+ stores — creating a potential competition between DNA repair and other NAD+-dependent processes.
- PARP overactivation: Chronic or excessive PARP activation — as may occur with accumulating DNA damage in aging models — has been studied as a driver of progressive NAD+ depletion, creating a feedback loop where diminished NAD+ further impairs DNA repair capacity.
- CD38 as a major NAD+ consumer: CD38 is an ectoenzyme that hydrolyzes NAD+ to produce cyclic ADP-ribose (cADPR) and ADPR — calcium-signaling molecules. Research documents that CD38 expression and activity increase with age and inflammation, contributing significantly to age-associated NAD+ decline.
- CD38 inhibition research: Preclinical models exploring CD38 inhibitors in combination with NAD+ precursor supplementation have shown measurable effects on tissue NAD+ levels and metabolic markers, representing an active area of aging biology investigation.
Cognitive Research and Neurological Models
The brain is among the highest-energy-consuming organs in the body, and NAD+-dependent processes are correspondingly active in neural tissue. Research in neurological models has examined NAD+’s role in both neuroprotective signaling and neuroinflammatory regulation.
Documented research findings include:
- Neuroprotective signaling: Preclinical models demonstrate NAD+-dependent neuroprotective signaling, including SIRT1-mediated protection against neuronal apoptosis and PARP-1-dependent DNA repair in neural cells following oxidative stress exposure.
- Neuroinflammation modulation: In vitro studies have documented NAD+ involvement in NF-κB pathway modulation — a key regulator of neuroinflammatory gene expression — with NAD+ repletion associated with attenuated inflammatory signaling in relevant cell models.
- Aging brain NAD+ depletion: Animal models document progressive NAD+ depletion in aging brain tissue, associated with declining SIRT1 activity and increased markers of oxidative stress and neuroinflammation.
- Combination research: Researchers studying nootropic peptides and NAD+ in combination models — including Semax, a neuropeptide studied for its neurotrophic and neuroprotective properties — represent an emerging area of preclinical investigation into multi-mechanism cognitive aging models.
NAD+ Injectable vs. Oral Research Models
Research designs involving NAD+ must account for significant differences in delivery route pharmacokinetics. The choice between injectable and oral models substantially affects plasma concentration curves, tissue distribution, and experimental design validity.
- Injectable NAD+ (IV or IM): Provides the fastest and most direct plasma concentration increases in research settings. Bypasses gastrointestinal metabolism entirely. Used in research contexts where rapid tissue repletion or precise concentration control is required.
- Oral NAD+: Has poor bioavailability — NAD+ itself is poorly absorbed across the intestinal epithelium and is largely hydrolyzed before absorption. Oral NAD+ supplementation research has largely shifted to precursor molecules for this reason.
- Oral precursors (NMN, NR): Studied as oral alternatives that can be absorbed and enzymatically converted to NAD+ intracellularly. Each has distinct absorption, conversion efficiency, and tissue distribution profiles.
- Research design selection: Injectable models are typically chosen when direct NAD+ repletion data is needed without the confound of precursor conversion kinetics. Oral precursor models are selected when gut absorption and systemic distribution are part of the research question.
Researchers sourcing injectable NAD+ for research protocols can review Spartan Peptides’ NAD+ research compound — available as lyophilized powder for reconstitution.
NAD+ vs. NMN vs. NR: What Research Compares
Research comparing NAD+ and its primary precursors — NMN (Nicotinamide Mononucleotide) and NR (Nicotinamide Riboside) — has grown substantially, with each form showing distinct pharmacokinetic profiles and tissue outcomes in preclinical models.
- NAD+ (direct form): The active coenzyme form. Injectable delivery provides immediate plasma repletion without enzymatic conversion steps. Preferred in research designs requiring controlled and rapid tissue-level NAD+ increases.
- NMN (Nicotinamide Mononucleotide): A direct precursor one step upstream of NAD+. Requires conversion by NMNAT enzymes (nicotinamide mononucleotide adenylyltransferases) inside cells. Studied extensively as an oral and injectable supplement in mouse aging models — including the landmark studies from the Imai laboratory at Washington University.
- NR (Nicotinamide Riboside): A precursor two steps upstream of NAD+. Converted first to NMN by NR kinases, then to NAD+ by NMNATs. Studied primarily as an oral supplement with demonstrated oral bioavailability in multiple clinical and preclinical studies.
Research comparing forms typically evaluates:
- Speed of plasma and tissue NAD+ repletion
- Tissue-specific distribution and uptake efficiency
- Stability under storage and reconstitution conditions
- Cost-per-research-dose relative to achievable tissue concentrations
Injectable NAD+ — available from Spartan Peptides as a research-grade NAD+ compound — is the form of choice when direct repletion without precursor conversion is the research objective.
Sourcing NAD+ for Research
Research validity depends on compound quality. For NAD+, several sourcing parameters are critical to any rigorous experimental design:
- Purity: ≥98% HPLC verification — Lower purity preparations frequently contain degradation products including ADPR (ADP-ribose) and AMP, which have their own biological activities and can confound results. Third-party HPLC-verified purity documentation should accompany every research-grade NAD+ batch.
- Lyophilized powder form: NAD+ in lyophilized (freeze-dried) powder form offers significantly greater stability during storage and shipping compared to liquid preparations. Reconstitution with bacteriostatic water prior to use is standard practice in research protocols.
- Certificate of Analysis (CoA): A batch-specific, third-party CoA confirming identity, purity, and absence of contaminants is a non-negotiable requirement for research use. Each CoA should specify HPLC purity percentage, molecular weight confirmation, and residual solvent testing where applicable.
- USA-manufactured supply chain: Domestic manufacturing provides regulatory traceability and supply chain consistency. Research institutions often require domestic sourcing for compliance purposes.
- Packaging integrity: Hermetically sealed, nitrogen-purged vials minimize moisture and oxidation exposure during storage — both of which degrade NAD+ stability.
Spartan Peptides’ NAD+ research compound is HPLC-verified at ≥98% purity, supplied as lyophilized powder, USA manufactured, and available with third-party CoA documentation for research use.
Frequently Asked Questions
Is NAD+ a peptide?
Technically, NAD+ (Nicotinamide Adenine Dinucleotide) is a dinucleotide coenzyme, not a peptide. Peptides are chains of amino acids; NAD+ is a nucleotide-derived molecule. However, NAD+ is commonly grouped with research peptides because it is used in the same longevity and cellular optimization research contexts, often alongside peptides like BPC-157, GHK-Cu, and MOTS-c. Many research suppliers carry NAD+ in their peptide catalog for this reason.
What does NAD+ do in cells?
NAD+ functions as an essential coenzyme in cellular energy metabolism. It serves as an electron carrier in mitochondrial oxidative phosphorylation, a substrate for sirtuin deacetylases (SIRT1–7) involved in gene regulation, and a substrate for PARP enzymes in DNA strand break repair. NAD+ is present in every living cell and participates in hundreds of metabolic reactions.
Why does NAD+ decline with age?
Research documents that NAD+ levels decline with age due to increased consumption by CD38 (an ectoenzyme upregulated with age and inflammation) and PARP (activated by accumulating DNA damage). Simultaneously, salvage pathway efficiency decreases. The result is a documented reduction in NAD+ availability in tissues studied in aging models.
What is the difference between NAD+ and NMN?
NAD+ is the active coenzyme form; NMN (Nicotinamide Mononucleotide) is a biosynthetic precursor that must be converted to NAD+ inside cells. Injectable NAD+ provides direct repletion without the conversion step. NMN is studied primarily as an oral supplement with different pharmacokinetics. Research designs typically choose between them based on desired delivery route and speed of tissue repletion.
What purity is required for research-grade NAD+?
Research-grade NAD+ requires ≥98% HPLC-verified purity with third-party certificate of analysis documentation. Degradation products (ADPR, AMP) can be present in lower-purity preparations and interfere with research validity. Lyophilized powder form provides the best stability during storage.
Where can researchers source NAD+ for research use?
Research-grade NAD+ should be sourced from a supplier offering ≥98% HPLC verification, third-party CoA, and domestic (USA) manufacturing. Spartan Peptides offers injectable NAD+ for research use with full documentation.
NAD+’s position as a central regulator of mitochondrial function, sirtuin signaling, and DNA repair has made it one of the most actively researched compounds in longevity science. Whether used alone or in combination with other research peptides, the quality of the NAD+ compound directly affects research validity. Spartan Peptides’ NAD+ research compound is HPLC-verified at ≥98% purity, USA manufactured, and available for research use.
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease. For research use only. Not for human consumption.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →