Complete Guide to Anti-Aging Peptides
Written bySpartan Research Team
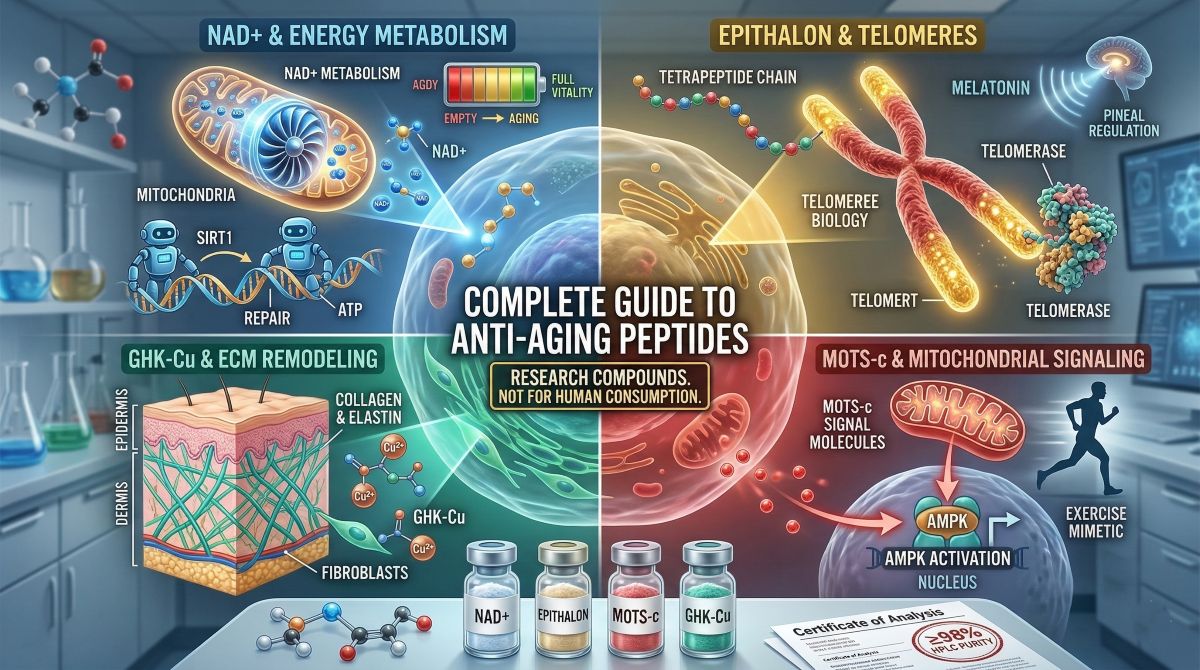
Anti-aging peptides occupy one of the most scientifically active areas of longevity research. The growing body of work on cellular aging has moved the field well past the descriptive — cataloguing what changes with age — toward the mechanistic: identifying which molecular processes drive senescence, genomic instability, and metabolic decline, and whether specific compounds can modulate those processes. Peptides have become central to this effort because they operate as precise molecular signals within the biological systems that govern aging, rather than producing broad pharmacological effects that are difficult to interpret or translate.
This guide covers the primary anti-aging and longevity peptides currently under scientific investigation. For each compound, it addresses the molecular biology it targets, the specific mechanisms through which it acts, and the experimental evidence supporting its research interest. The compounds covered — NAD+, Epithalon, MOTS-c, and GHK-Cu — represent distinct classes targeting different rate-limiting processes in cellular aging: NAD+ metabolism, telomere biology, mitochondrial regulation, and extracellular matrix maintenance. Each mechanism is relevant on its own; together, they map the current frontier of peptide-based aging research.
All compounds discussed here are research peptides supplied strictly for laboratory investigation. They are not approved for human consumption and are referenced exclusively in the context of scientific research.
The Molecular Biology of Aging: What Anti-Aging Peptides Target
Aging is not a single process. It is the cumulative result of several parallel molecular deterioration programs that interact with each other and with environmental stressors over time. The research peptides in this category each address one or more of these programs. The primary biological processes targeted by current longevity peptide research are:
NAD+ Depletion and Energy Metabolism
Nicotinamide adenine dinucleotide (NAD+) is a coenzyme present in every cell, where it functions as an electron carrier in oxidative phosphorylation and as a substrate for enzymes involved in DNA repair (PARPs), gene expression regulation (sirtuins), and cellular stress response (CD38). NAD+ levels decline systematically with age — typically 50% or more between young adulthood and middle age — and this depletion impairs the activity of all NAD+-dependent enzymes simultaneously. The consequence is a broad deterioration in mitochondrial function, DNA repair capacity, and metabolic flexibility that is increasingly recognized as a convergence point for multiple aging phenotypes.
Telomere Shortening and Chromosomal Instability
Telomeres are repetitive DNA sequences (TTAGGG in mammals) that cap chromosome ends and protect them from degradation and end-joining events. With each cell division, telomeres shorten by 50–200 base pairs due to the end-replication problem — the inability of DNA polymerase to fully replicate the 3′ end of linear chromosomes. When telomere length falls below a threshold, the cell activates DNA damage response pathways and enters replicative senescence or apoptosis. Telomere attrition rate correlates with biological age across multiple human studies, and telomere length is one of the most studied biomarkers of cellular aging. Telomerase — the enzyme that extends telomeres — is expressed in stem cells and germline cells but is largely silenced in somatic cells, making its reactivation a target of active research.
Mitochondrial Dysfunction
Mitochondria generate most of the cell’s ATP through oxidative phosphorylation, and their efficiency declines with age through a combination of mitochondrial DNA (mtDNA) mutation accumulation, reduced mitochondrial biogenesis, impaired mitophagy (selective clearance of damaged mitochondria), and decreased membrane potential. These changes reduce energy production, increase reactive oxygen species (ROS) generation, and impair the cell’s capacity to adapt to metabolic demands. In post-mitotic tissues — neurons, cardiomyocytes, skeletal muscle fibers — mitochondrial decline is particularly consequential because these cells cannot dilute damaged mitochondria through division.
Extracellular Matrix Degradation
The extracellular matrix (ECM) provides the structural scaffold within which cells reside and through which they communicate via growth factor binding and mechanotransduction. Aging ECM is characterized by collagen cross-link accumulation (leading to tissue stiffening), matrix metalloproteinase (MMP) upregulation (leading to ECM degradation), and reduced synthesis of new structural proteins. Skin aging is the most visible manifestation of ECM deterioration, but the same processes affect vascular walls, joint cartilage, and organ parenchyma. Peptide-based interventions targeting ECM maintenance — particularly copper peptides — are an active area of both cosmetic and systemic aging research.
| ★ | Research context: The Hallmarks of Aging framework (Lopez-Otin et al., 2013, updated 2023) identifies twelve interconnected biological processes driving aging, including genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis. The research compounds covered in this guide target at least four of these hallmarks directly. |
Anti-Aging Peptides: Compound Profiles

- NAD+ (Nicotinamide Adenine Dinucleotide)
NAD+ is technically a dinucleotide rather than a peptide, but its inclusion in longevity research compound catalogs reflects its central position in the biochemistry of aging. As a cofactor and signaling molecule, NAD+ connects energy metabolism (via the electron transport chain), genome maintenance (via PARP1/2 activity), and epigenetic regulation (via sirtuin deacylase activity) in a single metabolic network. Age-related NAD+ depletion, therefore, has consequences across all three of these processes simultaneously, which is why its restoration has attracted considerable research attention as a broad-spectrum approach to attenuating cellular aging. The complete research overview is available in our NAD+ 750mg Complete Research Guide.
NAD+ Mechanism: Sirtuins, PARPs, and Metabolic Regulation
Sirtuins (SIRT1–7) are NAD+-dependent deacylases that regulate gene expression through histone modification, transcription factor activity, and mitochondrial biogenesis. SIRT1 deacetylates PGC-1alpha — the master regulator of mitochondrial biogenesis — promoting mitochondrial turnover and energy metabolism. SIRT3 maintains mitochondrial function by deacetylating electron transport chain components and antioxidant enzymes. SIRT6 regulates DNA repair and telomere maintenance. All sirtuin activity is strictly NAD+-dependent, so as NAD+ levels fall with age, sirtuin activity falls proportionally, impairing all of these downstream processes.
PARP1 and PARP2 are DNA repair enzymes that consume NAD+ as a substrate during single-strand break repair. In aged or oxidatively stressed cells, elevated DNA damage levels drive excessive PARP activation, which accelerates NAD+ depletion and creates a feedback loop: more DNA damage → more PARP activity → less NAD+ → less sirtuin activity → less mitochondrial quality control → more ROS → more DNA damage.
CD38 is an NAD+-consuming enzyme whose expression increases with age and in senescent cells, contributing further to the age-related NAD+ decline independently of PARP activity.
NAD+ Research Evidence
The foundational preclinical work from the Sinclair laboratory at Harvard (Gomes et al., 2013, Cell) demonstrated that NMN supplementation in aged mice restored NAD+ levels and improved mitochondrial function to levels comparable to those of younger animals within one week. Subsequent studies have documented NAD+ repletion improving insulin sensitivity in obese mice (Yoshino et al., 2011), extending lifespan in C. elegans, and attenuating age-related muscle decline in rodent models. Human pharmacokinetic studies have confirmed that orally administered NAD+ precursors (NMN, NR) dose-dependently elevate blood NAD+ levels (Trammell et al., 2016; Airhart et al., 2017). For a direct comparison of NAD+ precursor compounds, see NAD+ vs. NMN vs. NR: Precursor Comparison Research.
Spartan Peptides supplies NAD+ research compound at ≥98% HPLC-verified purity. For an overview of what NAD+ does at the cellular level, see What Does NAD+ Do?.
- Epithalon (Epitalon)
Epithalon (also written Epitalon) is a synthetic tetrapeptide (Ala-Glu-Asp-Gly) developed by Vladimir Khavinson and colleagues at the St. Petersburg Institute of Bioregulation and Gerontology. It was synthesized as an analogue of epithalamin, a naturally occurring polypeptide extract from the pineal gland, and has been studied primarily for its effects on telomere biology and pineal gland function. Among anti-aging peptides, Epithalon has one of the most extensive research histories in terms of longevity endpoints, with animal lifespan studies and human observational data published over several decades. The mechanistic and clinical data are reviewed in our blog Can We Reverse Aging?.
Epithalon Mechanism: Telomerase Activation and Pineal Regulation
Epithalon’s two primary documented mechanisms are telomerase activation and regulation of melatonin synthesis through the pineal gland. On telomere biology: in vitro studies using human somatic cells have shown that Epithalon activates telomerase (hTERT) expression, enabling telomere elongation in cell types that normally lack telomerase activity. This is mechanistically relevant because telomerase reactivation in somatic cells is a longstanding theoretical target for extending replicative lifespan — the number of cell divisions a cell can undergo before entering senescence.
On pineal regulation: Epithalon has been shown in animal models to restore age-related decline in pineal melatonin synthesis. Melatonin is itself an antioxidant and circadian regulator, and its age-related decline contributes to disrupted sleep architecture, increased oxidative stress, and impaired immune surveillance — all processes associated with accelerated aging phenotypes. The mechanism involves Epithalon’s effect on the expression of hydroxyindole-O-methyltransferase (HIOMT), the enzyme catalyzing the final step in melatonin biosynthesis.
Epithalon Research Evidence
Khavinson et al. (2003) reported that Epithalon stimulated telomerase activity in human fetal fibroblasts and extended cell division capacity beyond the normal Hayflick limit. This in vitro finding provided the mechanistic basis for Epithalon’s positioning as a telomere research compound. Anisimov et al. conducted a series of murine lifespan studies across different mouse strains, with consistent findings of extended mean and maximum lifespan in Epithalon-treated cohorts relative to controls. In a 25-year observational study in humans (Khavinson & Morozov, 2003), elderly subjects receiving periodic peptide bioregulator treatments, including Epithalon, showed reduced mortality rates and better functional status relative to control groups — though this observational design limits causal interpretation.
Additional research data are covered in Unlocking Longevity.
- MOTS-c
MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a 16-amino acid peptide encoded within the mitochondrial genome — specifically from the 12S ribosomal RNA gene — making it one of the very few peptides produced from mitochondrial rather than nuclear DNA.
This origin is biologically meaningful: MOTS-c functions as a retrograde signal from the mitochondria to the nucleus, communicating mitochondrial metabolic status and triggering adaptive gene expression in response to energetic stress.
In aging research, it has attracted attention for its capacity to improve metabolic function, attenuate muscle decline, and act as an exercise mimetic in aged animal models.
MOTS-c Mechanism: AMPK Activation and the Folate Cycle
The primary downstream target of MOTS-c is AMP-activated protein kinase (AMPK) — the cell’s master energy sensor. AMPK is activated when the AMP: ATP ratio rises (indicating low energy availability), and it triggers a program of cellular adaptations: increased fatty acid oxidation, enhanced glucose uptake, mitochondrial biogenesis, and autophagy induction. In aged cells, AMPK activation becomes blunted — a change associated with accumulation of lipid in non-adipose tissues, insulin resistance, and reduced mitochondrial turnover.
Mechanistically, MOTS-c reaches the nucleus under metabolic stress and regulates the folate cycle and de novo purine biosynthesis. This regulation affects AICAR (5-aminoimidazole-4-carboxamide ribonucleotide) levels, an endogenous AMPK agonist. By raising intracellular AICAR, MOTS-c activates AMPK without requiring exogenous exercise-derived AMP elevation — hence its classification as an exercise mimetic in the literature.
A second, age-specific mechanism was documented by Kim et al. (2022, Nature Aging): circulating MOTS-c levels decline with age in both mice and humans, and exogenous MOTS-c administration in aged male mice improved physical performance, metabolic parameters, and survival rates. The study also found that MOTS-c acts in the skeletal muscle to regulate gene networks associated with proteostasis and stress response — pathways central to muscle aging.
- GHK-Cu (Copper Peptide)
GHK-Cu (glycyl-L-histidyl-L-lysine: copper 2+) is a naturally occurring copper complex identified in human plasma, urine, and saliva. It was first isolated by Loren Pickart in 1973 from human albumin and characterized for its stimulatory effects on liver tissue growth. Subsequent research across four decades has documented GHK-Cu’s effects on collagen synthesis, matrix metalloproteinase regulation, antioxidant gene expression, and DNA repair. In the anti-aging context, GHK-Cu addresses ECM deterioration and oxidative stress — two processes whose combined effect on tissue architecture accelerates with age.
GHK-Cu Mechanism: ECM Remodeling and Gene Expression
GHK-Cu operates through two primary mechanisms. First, it stimulates collagen synthesis in fibroblasts through upregulation of collagen gene expression, while simultaneously regulating MMP activity — promoting the degradation of damaged, cross-linked collagen and its replacement with newly synthesized matrix. This dual effect addresses both the accumulation of dysfunctional ECM components (a feature of aged tissue) and the synthesis deficit that prevents their replacement.
Second, Pickart and colleagues documented that GHK-Cu acts as a broad gene expression regulator, modulating hundreds of genes in directions consistent with tissue remodeling and anti-aging biology. Microarray analyses of fibroblasts treated with GHK-Cu showed upregulation of genes involved in antioxidant defense (superoxide dismutase, catalase), DNA repair, and proteoglycan synthesis, alongside downregulation of inflammatory cytokines and oncogenesis-associated genes. This pattern suggests GHK-Cu affects aging-relevant gene networks rather than a single pathway.
At the copper coordination level, the His-Gly-Lys backbone binds Cu2+ with high affinity in a square-planar configuration. The copper chelation is functionally important: copper is required for lysyl oxidase activity (which cross-links newly synthesized collagen and elastin), superoxide dismutase function, and cytochrome c oxidase activity in the mitochondrial electron transport chain.
Comparative Overview: Anti-Aging Peptides at a Glance
| Compound | Primary Target | Key Research Finding | Biological Process | Class |
|---|---|---|---|---|
| NAD+ | Sirtuin/PARP activation; mitochondrial energy metabolism | NMN repletion restores mitochondrial function in aged mice to young-animal levels (Gomes et al., 2013) | NAD+ metabolism, DNA repair, epigenetic regulation | Coenzyme/metabolite |
| Epithalon | Telomerase (hTERT) activation; pineal melatonin regulation | Extended mean and maximum lifespan in multiple murine strains; telomere elongation in human somatic cells | Telomere biology, replicative senescence | Tetrapeptide |
| MOTS-c | AMPK activation via the AICAR pathway; nuclear metabolic signaling | Improved physical performance, metabolic parameters, and survival in aged male mice (Kim et al., 2022) | Mitochondrial-nuclear signaling, metabolic flexibility | Mitochondria-derived peptide |
| GHK-Cu | Collagen synthesis, MMP regulation, antioxidant gene expression | Broad gene expression modulation in anti-aging directions; collagen and elastin synthesis stimulation | ECM remodeling, oxidative stress response | Copper peptide complex |
NAD+ Depletion as a Driver of Cellular Aging: The Evidence
The Age-Dependent Decline in NAD+
Multiple human tissue studies have now documented the magnitude and timeline of NAD+ decline with age. Massudi et al. (2012) analyzed NAD+ levels in human muscle biopsies and blood samples across age groups, finding a 50% reduction in NAD+ between subjects in their 20s and 50s. Yoshino et al. (2021) conducted the first randomized, placebo-controlled human trial of NMN supplementation (250mg/day for 10 weeks in postmenopausal women with prediabetes), demonstrating improved muscle insulin sensitivity and gene expression changes in skeletal muscle consistent with enhanced oxidative metabolism.
The NAD+ precursor research has been further stratified by route of synthesis. NAD+ is synthesized from tryptophan via the de novo pathway, from nicotinic acid via the Preiss-Handler pathway, and from nicotinamide, NMN, and NR via the salvage pathway. The salvage pathway predominates in most tissues and is the target of most NAD+-boosting interventions. Tissue distribution of the enzymes governing each precursor’s conversion differs, which means different precursors may be more effective in different organs — a research question that is actively studied.
Sirtuin Biology and Aging
SIRT1 has been the most extensively studied sirtuin in the aging context. In addition to deacetylating PGC-1alpha to promote mitochondrial biogenesis, SIRT1 deacetylates p53 (reducing apoptosis), NF-kB (reducing inflammation), and FOXO transcription factors (promoting stress resistance and autophagy). The breadth of SIRT1’s regulatory targets means that NAD+ depletion — by reducing SIRT1 activity — simultaneously impairs mitochondrial function, increases inflammatory signaling, reduces stress response capacity, and impairs autophagic clearance of cellular debris.
SIRT6 is specifically relevant to the DNA repair and telomere maintenance functions of sirtuins. It deacetylates histone H3K9Ac at telomeres, maintaining their heterochromatic state and reducing genomic instability. SIRT6 knockout mice age prematurely, while SIRT6 overexpression extends lifespan in male mice (Kanfi et al., 2012) — making it one of the most directly validated lifespan-regulatory genes identified in mammals.
Telomere Biology and Anti-Aging Peptide Research
Telomere Length as a Biomarker of Biological Age
The correlation between telomere length and biological aging has been established across multiple human cohort studies. Blackburn, Greider, and Szostak’s foundational telomere and telomerase work — for which they received the 2009 Nobel Prize in Physiology or Medicine — established the molecular basis for why telomere attrition drives replicative senescence. Subsequent epidemiological work has documented associations between shorter leukocyte telomere length and increased risk of cardiovascular disease, cancer, and all-cause mortality, though the causal interpretation of these associations remains an area of active research.
The Hayflick limit — the finite number of cell divisions a somatic cell can undergo before entering senescence — is directly tied to telomere length reaching a critical threshold. In the absence of telomerase activity, each division removes 50–200 base pairs, and after approximately 40–60 divisions (varying by cell type), the shortest telomeres trigger a DNA damage response that halts proliferation. Stem cells and germline cells express telomerase constitutively and can bypass this limit; somatic cells largely cannot.
Epithalon and Telomerase: What the Research Shows
Epithalon’s telomerase-activating effect, first documented by Khavinson et al. (2003) in human fetal fibroblasts and intestinal cells, has been reproduced in several subsequent in vitro studies. The mechanism is attributed to Epithalon’s effect on hTERT transcription — the catalytic subunit of telomerase that is the rate-limiting factor for telomerase activity in most somatic cells. Whether this in vitro finding translates to meaningful telomere length maintenance in vivo, and on what timescale, remains an open research question. The 2022 update of the Hallmarks of Aging framework identifies telomere attrition as one of the primary drivers of aging, which positions telomerase research compounds as a theoretically sound, if still early-stage, research area.
Combination Research: Targeting Multiple Aging Pathways

The multi-hallmark character of aging creates a research rationale for combining compounds that address different rate-limiting processes. NAD+ repletion, telomere maintenance, mitochondrial signaling, and ECM repair each represent a separate biological deficit that worsens with age; addressing one while leaving others unattended leaves substantial aging biology unaddressed. The anti-aging peptide stack research covers the current thinking on multi-compound longevity research protocols.
NAD+ and MOTS-c are particularly well-suited to co-investigation because their downstream targets converge on mitochondrial function from different upstream entry points. NAD+ acts through sirtuin-mediated transcriptional regulation and PARP1-dependent DNA repair; MOTS-c acts through AMPK activation and nuclear gene regulation. Both ultimately improve mitochondrial efficiency and reduce ROS production, but through mechanisms that are parallel rather than redundant — meaning each contributes an independent effect even when combined.
Epithalon addresses a mechanistically distinct aging process — telomere maintenance — that neither NAD+ repletion nor MOTS-c directly addresses. Adding a telomere-targeting compound to a mitochondria-focused protocol addresses the chromosomal stability axis of aging that would otherwise remain unaddressed. This complementarity is the research logic behind multi-compound anti-aging protocols.
For research that combines NAD+, Semax, and CJC-1295 across energy, cognitive, and GH axis targets, see Biohacking Research.
Research Evidence: Synthesis Across Compound Classes
Strength of Evidence by Compound
The anti-aging peptide evidence base varies considerably in quality and depth across the four compounds reviewed here. NAD+ has the strongest and most mechanistically characterized evidence, with human randomized controlled trial data (Yoshino et al., 2021; Trammell et al., 2016), extensive preclinical mechanistic studies, and multiple independent research groups confirming foundational findings. The translational path from preclinical to human data is more advanced for NAD+ than for any other compound in this group.
Epithalon has a long research history from a single primary laboratory (Khavinson, St. Petersburg), which provides both depth of data and a degree of caution about independent replication. The human observational studies are methodologically weaker than controlled trials, and the in vitro telomerase findings, while consistent, have not been reproduced in long-term controlled human studies. The compound’s research interest remains high because telomere biology is a well-validated aging mechanism and few other research tools target it directly.
MOTS-c has a shorter research history but higher-quality individual studies. The founding paper (Lee et al., 2015, Cell Metabolism) is from a high-impact journal with rigorous methodology, and the 2022 Nature Aging paper provides additional human-relevance data. Independent replication is growing, which is the marker of a maturing research area rather than a single laboratory’s findings.
GHK-Cu has a long research history in dermatological and wound healing contexts, with consistent in vitro data on collagen synthesis and gene expression. Its human data are predominantly from topical cosmetic research (skin texture, wound healing) rather than systemic aging biology, which limits the scope of claims about its systemic anti-aging effects.
Research-Grade Quality Standards for Anti-Aging Compounds
Anti-aging research involves long-duration protocols and subtle biological endpoints — changes in gene expression, telomere length, mitochondrial efficiency — that are highly sensitive to compound quality variation. A contaminated or degraded compound does not merely produce a smaller effect; it produces an uninterpretable result.
The quality requirements for anti-aging research compounds are:
- HPLC purity ≥98%: For peptides like Epithalon (a tetrapeptide, MW ~441 Da) and MOTS-c (a 16-mer, MW ~2174 Da), HPLC analysis at 214–220nm is the standard method for confirming that the target compound accounts for ≥98% of UV-absorbing material. Impurities at the sub-2% level are generally tolerated in research settings; higher impurity levels compromise dose-response reliability.
- Mass spectrometry confirmation: ESI-MS or MALDI-TOF verifying correct molecular weight is especially important for longer peptides like MOTS-c, where synthesis errors, including amino acid deletions, racemization, or incomplete deprotection, can produce a compound with correct mass minus one or two residues — an error that HPLC alone may not detect.
- Certificate of Analysis (CoA): Batch-specific documentation of purity data, mass spectra, and synthesis date. Required for any research protocol that needs to document compound provenance.
- Storage and stability: Lyophilized peptide powders are generally stable at -20°C but degrade at ambient temperatures over timescales relevant to shipping. For NAD+, oxidation during storage is a specific concern: NAD+ (oxidized form) and NADH (reduced form) have different biological activities, and a partially reduced supply introduces a confound. Cold-chain documentation should be requested from any research compound supplier.
Spartan Peptides applies ≥98% HPLC purity standards and provides batch-specific CoA documentation for all anti-aging and longevity research compounds. The full anti-aging and cellular health research catalog covers NAD+, Epithalon, and MOTS-c. For a research-oriented guide to evaluating peptide quality, see Quality Control: Interpreting Purity in Peptide Research.
Frequently Asked Questions
Q: Why do NAD+ levels fall with age, and why does this matter for anti-aging research?
NAD+ decline with age results from a combination of reduced biosynthesis and increased consumption. CD38 — an NAD+-consuming enzyme expressed by immune cells — increases with age and with the accumulation of senescent cells (which stimulate CD38 expression in neighboring cells via the senescence-associated secretory phenotype, SASP). Simultaneously, the age-related decline in mitochondrial activity reduces the demand for NAD+ recycling, shifting the biosynthesis-consumption balance further toward depletion. The research consequence is that multiple NAD+-dependent processes — PARP-mediated DNA repair, sirtuin-mediated gene regulation, mitochondrial quality control — are all impaired simultaneously. NAD+ repletion research, therefore, addresses multiple aging mechanisms through a single metabolic intervention, which is why it has attracted disproportionate scientific interest relative to single-pathway aging targets.
Q: What is the mechanistic basis for Epithalon’s effect on lifespan in animal studies?
Epithalon’s proposed lifespan-extending mechanism combines two parallel effects: telomerase activation in somatic cells, which extends replicative lifespan by slowing telomere attrition, and restoration of pineal melatonin synthesis, which improves circadian regulation and antioxidant defense. In the Anisimov murine studies, treated animals consistently showed extended mean and maximum lifespan relative to controls, and the treated animals maintained better circadian melatonin profiles and lower rates of spontaneous tumor formation. The mechanistic interpretation is that Epithalon addresses two independent aging rate-limiters — chromosomal stability and oxidative stress from circadian disruption — whose combined attenuation produces a measurable lifespan effect. The absence of human-controlled trial data limits how directly these animal findings can be translated, but the mechanistic basis is grounded in well-characterized aging biology.
Q: How does MOTS-c differ from other mitochondria-targeted research compounds?
MOTS-c is the only peptide in current research use that is encoded by mitochondrial DNA. This origin means it functions as a genuine retrograde signal from the mitochondria to the nucleus — a biological messenger that communicates mitochondrial status to nuclear gene expression machinery. Other mitochondria-targeted compounds (NAD+ precursors, coenzyme Q10, MitoQ) work by delivering substrates or antioxidants to the mitochondrial environment, or by improving electron transport chain efficiency. MOTS-c, by contrast, activates a gene regulatory program that restructures cellular metabolism from the nucleus — a more upstream intervention that addresses multiple downstream mitochondrial aging phenotypes through a single signaling event. Its status as an exercise mimetic — producing AMPK activation without exogenous exercise — positions it as a research tool for studying metabolic aging in sedentary models.
Q: Are anti-aging peptides the same as longevity supplements?
Research peptides for anti-aging are not supplements. They are research compounds supplied for laboratory investigation of aging biology, and they are not approved by the FDA for human consumption. Longevity supplements — such as resveratrol, nicotinamide riboside, or quercetin — are generally available as over-the-counter products with consumer labeling and varying regulatory status. The research compounds covered in this guide are held to higher purity standards (≥98% HPLC), require CoA documentation, and are supplied specifically for use in controlled research settings, not for consumer use. This distinction is both regulatory and practical: research-grade compounds require documented purity and analytical verification that consumer supplement manufacturing does not routinely provide.
Q: What quality parameters should a research protocol specify for anti-aging peptides?
For any anti-aging peptide research protocol, the minimum specifications should include: HPLC purity ≥98% with UV detection at 214–220nm; mass spectrometry confirmation of molecular weight within accepted error margins for the target compound; batch-specific CoA with synthesis date; and cold-chain documentation for shipping. For longer experiments involving repeated administration — which is common in lifespan research — batch-to-batch consistency documentation adds traceability. For MOTS-c specifically, given its mitochondrial-gene origin and the precision required for AMPK pathway research, mass spectrometry confirmation of the full 16-residue sequence is particularly warranted.
Conclusion
Anti-aging peptide research has matured considerably over the past decade, progressing from descriptive observations about age-associated changes to mechanistically grounded investigations of compounds that target specific molecular drivers of cellular aging. NAD+ addresses the coenzyme depletion that undermines sirtuin and PARP function simultaneously. Epithalon targets telomere biology through telomerase activation — one of the most direct interventions in replicative aging currently available as a research compound. MOTS-c operates at the mitochondria-to-nucleus signaling interface, restoring metabolic adaptability that declines with age. GHK-Cu addresses the ECM deterioration that alters tissue architecture and function throughout the body. Together, these compounds represent a multi-mechanism approach to investigating the biological processes that limit healthy lifespan.
Spartan Peptides supplies all four compounds — NAD+, Epithalon, MOTS-c, and GHK-Cu — at ≥98% HPLC-verified purity with full CoA documentation.
Disclaimer: All products offered by Spartan Peptides are intended for laboratory research purposes only. They are not approved by the FDA for human consumption, and are not intended to diagnose, treat, cure, or prevent any disease or medical condition. This content is provided for informational and educational purposes only and does not constitute medical advice.
References
Lopez-Otin, C. et al. (2013). The Hallmarks of Aging. Cell, 153(6), 1194–1217. Updated: Lopez-Otin et al. (2023). Hallmarks of Aging: An Expanding Universe. Cell, 186(2), 243–278.
Gomes, A.P. et al. (2013). Declining NAD+ Induces a Pseudohypoxic State Disrupting Nuclear-Mitochondrial Communication during Aging. Cell, 155(7), 1624–1638.
Yoshino, M. et al. (2021). Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science, 372(6547), 1224–1229.
Trammell, S.A. et al. (2016). Nicotinamide riboside is uniquely and orally bioavailable in healthy humans. Nature Communications, 7, 12948.
Kanfi, Y. et al. (2012). The sirtuin SIRT6 regulates lifespan in male mice. Nature, 483(7388), 218–221.
Khavinson, V.Kh. et al. (2003). Peptide Epitalon activates telomerase in human somatic cells. Neuro Endocrinology Letters, 24(3–4), 162–165.
Anisimov, V.N. et al. (2010). Effect of Epitalon on biomarkers of aging, life span and spontaneous tumor incidence in female Swiss-derived SHR mice. Biogerontology, 11(4), 461–472.
Lee, C. et al. (2015). The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metabolism, 21(3), 443–454.
Kim, S.J. et al. (2022). Mitochondrially derived peptides as novel regulators of metabolism. Journal of Physiology, 595(21), 6511–6519.
Pickart, L. & Margolina, A. (2018). Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. International Journal of Molecular Sciences, 19(7), 1987.
Written by the Spartan Research Team
Our team of peptide researchers and biochemists reviews every article for scientific accuracy. Learn more about our team →